Polycystin-2 immunolocalization and function in zebrafish
- PMID: 16943304
- PMCID: PMC3698611
- DOI: 10.1681/ASN.2006040412
Polycystin-2 immunolocalization and function in zebrafish
Abstract
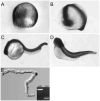
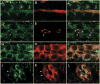
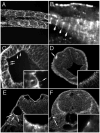
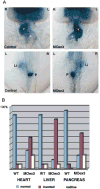
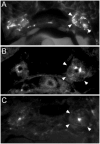
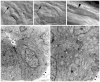
Polycystin-2 functions as a cation-permeable transient receptor potential ion channel in kidney epithelial cells and when mutated results in human autosomal dominant polycystic kidney disease. For further exploration of the in vivo functions of Polycystin-2, this study examined its expression and function during zebrafish embryogenesis. pkd2 mRNA is ubiquitously expressed, and its presence in the larval kidney could be confirmed by reverse transcription-PCR on isolated pronephroi. Immunostaining with anti-zebrafish Polycystin-2 antibody revealed protein expression in motile kidney epithelial cell cilia and intracellular cell membranes. Intracellular localization was segment specific; in the proximal nephron segment, Polycystin-2 was localized to basolateral cell membranes, whereas in the caudal pronephric segment, Polycystin-2 was concentrated in subapical cytoplasmic vesicles. Polycystin-2 also was expressed in muscle cells and in a variety of sensory cells that are associated with mechanotransduction, including cells of the ear, the lateral line organ, and the olfactory placodes. Disruption of Polycystin-2 mRNA expression resulted in pronephric kidney cysts, body axis curvature, organ laterality defects, and hydrocephalus-defects that could be rescued by expression of a human PKD2 mRNA. In-frame deletions in the first extracellular loop and C-terminal phosphofurin acidic cluster sorting protein-1 (PACS-1) binding sites in the cytoplasmic tail caused Polycystin-2 mislocalization to the apical cell surface. Unlike zebrafish intraflagellar transport protein (IFT) mutants, cyst formation was not associated with cilia defects and instead correlated with reduced kidney fluid output, expansion of caudal duct apical cell membranes, and occlusion of the caudal pronephric nephron segment.
Figures




Similar articles
-
The exocyst protein Sec10 interacts with Polycystin-2 and knockdown causes PKD-phenotypes.PLoS Genet. 2011 Apr;7(4):e1001361. doi: 10.1371/journal.pgen.1001361. Epub 2011 Apr 7. PLoS Genet. 2011. PMID: 21490950 Free PMC article.
-
Channel Function of Polycystin-2 in the Endoplasmic Reticulum Protects against Autosomal Dominant Polycystic Kidney Disease.J Am Soc Nephrol. 2022 Aug;33(8):1501-1516. doi: 10.1681/ASN.2022010053. Epub 2022 Jul 14. J Am Soc Nephrol. 2022. PMID: 35835458 Free PMC article.
-
Phosphodiesterase 1A modulates cystogenesis in zebrafish.J Am Soc Nephrol. 2014 Oct;25(10):2222-30. doi: 10.1681/ASN.2013040421. Epub 2014 Apr 3. J Am Soc Nephrol. 2014. PMID: 24700876 Free PMC article.
-
Regulation of polycystin expression, maturation and trafficking.Cell Signal. 2020 Aug;72:109630. doi: 10.1016/j.cellsig.2020.109630. Epub 2020 Apr 8. Cell Signal. 2020. PMID: 32275942 Free PMC article. Review.
-
Polycystic kidney disease: novel insights into polycystin function.Trends Mol Med. 2023 Apr;29(4):268-281. doi: 10.1016/j.molmed.2023.01.005. Epub 2023 Feb 15. Trends Mol Med. 2023. PMID: 36805211 Review.
Cited by
-
Hemodynamic Forces Regulate Cardiac Regeneration-Responsive Enhancer Activity during Ventricle Regeneration.Int J Mol Sci. 2021 Apr 11;22(8):3945. doi: 10.3390/ijms22083945. Int J Mol Sci. 2021. PMID: 33920448 Free PMC article.
-
Zebrafish as a Model for Drug Screening in Genetic Kidney Diseases.Front Pediatr. 2018 Jun 28;6:183. doi: 10.3389/fped.2018.00183. eCollection 2018. Front Pediatr. 2018. PMID: 30003073 Free PMC article. Review.
-
Zebrafish assays of ciliopathies.Methods Cell Biol. 2011;105:257-72. doi: 10.1016/B978-0-12-381320-6.00011-4. Methods Cell Biol. 2011. PMID: 21951534 Free PMC article.
-
Cilia functions in development.Curr Opin Cell Biol. 2012 Feb;24(1):24-30. doi: 10.1016/j.ceb.2011.12.007. Epub 2012 Jan 4. Curr Opin Cell Biol. 2012. PMID: 22226236 Free PMC article. Review.
-
Nephrocystin-3 is required for ciliary function in zebrafish embryos.Am J Physiol Renal Physiol. 2010 Jul;299(1):F55-62. doi: 10.1152/ajprenal.00043.2010. Epub 2010 May 12. Am J Physiol Renal Physiol. 2010. PMID: 20462968 Free PMC article.
References
-
- Clapham DE. TRP channels as cellular sensors. Nature. 2003;426:517–524. - PubMed
-
- Cai Y, Maeda Y, Cedzich A, Torres VE, Wu G, Hayashi T, Mochizuki T, Park JH, Witzgall R, Somlo S. Identification and characterization of polycystin-2, the PKD2 gene product. J Biol Chem. 1999;274:28557–28565. - PubMed
-
- Foggensteiner L, Bevan AP, Thomas R, Coleman N, Boulter C, Bradley J, Ibraghimov-Beskrovnaya O, Klinger K, Sandford R. Cellular and subcellular distribution of polycystin-2, the protein product of the PKD2 gene. J Am Soc Nephrol. 2000;11:814–827. - PubMed
-
- Koulen P, Cai Y, Geng L, Maeda Y, Nishimura S, Witzgall R, Ehrlich BE, Somlo S. Polycystin-2 is an intracellular calcium release channel. Nat Cell Biol. 2002;4:191–197. - PubMed
-
- Nauli SM, Alenghat FJ, Luo Y, Williams E, Vassilev P, Li X, Elia AE, Lu W, Brown EM, Quinn SJ, Ingber DE, Zhou J. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet. 2003;33:129–137. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

