Sonoporation from jetting cavitation bubbles
- PMID: 16950843
- PMCID: PMC1635670
- DOI: 10.1529/biophysj.105.075366
Sonoporation from jetting cavitation bubbles
Abstract
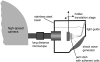
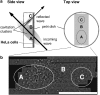
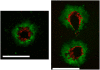
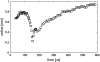
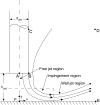
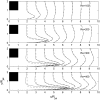
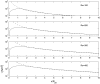
The fluid dynamic interaction of cavitation bubbles with adherent cells on a substrate is experimentally investigated. We find that the nonspherical collapse of bubbles near to the boundary is responsible for cell detachment. High-speed photography reveals that a wall bounded flow leads to the detachment of cells. Cells at the edge of the circular area of detachment are found to be permanently porated, whereas cells at some distance from the detachment area undergo viable cell membrane poration (sonoporation). The wall flow field leading to cell detachment is modeled with a self-similar solution for a wall jet, together with a kinetic ansatz of adhesive bond rupture. The self-similar solution for the delta-type wall jet compares very well with the full solution of the Navier-Stokes equation for a jet of finite thickness. Apart from annular sites of sonoporation we also find more homogenous patterns of molecule delivery with no cell detachment.
Figures






References
-
- Miller, M. W., D. L. Miller, and A. A. Brayman. 1996. A review of in vitro bioeffects of inertial ultrasonic cavitation from a mechanistic perspective. Ultrasound Med. Biol. 22:1131–1154. - PubMed
-
- Ng, K., and Y. Liu. 2002. Therapeutic ultrasound: its application in drug delivery. Med. Res. Rev. 22:204–223. - PubMed
-
- Bao, S. P., B. D. Thrall, and D. L. Miller. 1997. Transfection of a reporter plasmid into cultured cells by sonoporation in vitro. Ultrasound Med. Biol. 23:953–959. - PubMed
-
- Ward, M., J. R. Wu, and J. F. Chiu. 1999. Ultrasound-induced cell lysis and sonoporation enhanced by contrast agents. J. Acoust. Soc. Am. 105:2951–2957. - PubMed
-
- Stride, E., and N. Saffari. 2003. Microbubble ultrasound contrast agents: a review. Proc. Instn. Mech. Engrs. 217:429–447. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

