Deciphering the spike train of a sensory neuron: counts and temporal patterns in the rat whisker pathway
- PMID: 16957078
- PMCID: PMC6674492
- DOI: 10.1523/JNEUROSCI.1491-06.2006
Deciphering the spike train of a sensory neuron: counts and temporal patterns in the rat whisker pathway
Erratum in
- J Neurosci. 2006 Sep 20;26(38):9835
Abstract
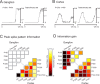
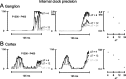
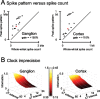
Rats achieve remarkable texture discriminations by sweeping their facial whiskers along surfaces. This work explores how neurons at two levels of the sensory pathway, trigeminal ganglion and barrel cortex, carry information about such stimuli. We identified two biologically plausible coding mechanisms, spike counts and patterns, and used "mutual information" to quantify how reliably neurons in anesthetized rats reported texture when "decoded" according to these candidate mechanisms. For discriminations between surfaces of different coarseness, spike counts could be decoded reliably and rapidly (within 30 ms after stimulus onset in cortex). Information increased as responses were considered as spike patterns with progressively finer temporal precision. At highest temporal resolution (spike sequences across six bins of 4 ms), the quantity of "information" in patterns rose 150% for ganglion neurons and 110% for cortical neurons above that in spike counts. In some cases, patterns permitted discriminations not supported by spike counts alone.
Figures





References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
