Isolation of high-affinity single-chain antibodies against Mycobacterium avium subsp. paratuberculosis surface proteins from sheep with Johne's disease
- PMID: 16960114
- PMCID: PMC1563570
- DOI: 10.1128/CVI.00163-06
Isolation of high-affinity single-chain antibodies against Mycobacterium avium subsp. paratuberculosis surface proteins from sheep with Johne's disease
Abstract
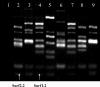
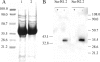
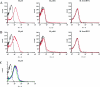
Johne's disease, caused by infection with Mycobacterium avium subsp. paratuberculosis, causes significant economic losses to the livestock farming industry. Improved investigative and diagnostic tools-necessary to understand disease processes and to identify subclinical infection-are much sought after. Here, we describe the production of single-chain antibodies with defined specificity for M. avium subsp. paratuberculosis surface proteins. Single-chain antibodies (scFv) were generated from sheep with Johne's disease by cloning heavy-chain and lambda light-chain variable regions and expressing these in fusion with gene III of filamentous phages. Two scFv clones (designated SurfS1.2 and SurfS2.2) were shown to be immunoreactive against M. avium subsp. paratuberculosis surface targets by flow cytometry, and immunoblotting identified specificity for a 34-kDa proteinase-susceptible determinant. Both antibodies were cross-reactive against Mycobacterium avium subsp. avium but nonreactive against Mycobacterium bovis or Mycobacterium phlei cells and were shown to be capable of enriching M. avium subsp. paratuberculosis cells by a factor of approximately 10(6)-fold when employed in magnetic bead separation of mixed Mycobacterium sp. cultures. Further, magnetic bead separation using SurfS1.2 and SurfS2.2 was capable of isolating as few as 10(3) M. avium subsp. paratuberculosis cells from ovine fecal samples, indicating the diagnostic potential of these reagents. Finally, inclusion of SurfS1.2 or SurfS2.2 in in vitro broth culture with M. avium subsp. paratuberculosis indicated that surface binding activity did not impede bacterial growth, although colony clumping was prevented. These results are discussed in terms of the potential use of single-chain phage display monoclonal antibodies as novel diagnostic reagents.
Figures

Similar articles
-
New method of serological testing for Mycobacterium avium subsp. paratuberculosis (Johne's disease) by flow cytometry.Foodborne Pathog Dis. 2005 Fall;2(3):250-62. doi: 10.1089/fpd.2005.2.250. Foodborne Pathog Dis. 2005. PMID: 16156706
-
PCR-restriction endonuclease analysis for identification and strain typing of Mycobacterium avium subsp. paratuberculosis and Mycobacterium avium subsp. avium based on polymorphisms in IS1311.Mol Cell Probes. 1999 Apr;13(2):115-26. doi: 10.1006/mcpr.1999.0227. Mol Cell Probes. 1999. PMID: 10208802
-
Development of ELISA to detect antibodies specific to Mycobacterium avium subsp. paratuberculosis with truncated 34 kDa proteins.Jpn J Vet Res. 2006 Nov;54(2-3):99-107. Jpn J Vet Res. 2006. PMID: 17201195
-
An evaluation of mycophage therapy, chemotherapy and vaccination for control of Mycobacterium avium subsp. paratuberculosis infection.Vet Microbiol. 2004 Dec 9;104(3-4):143-55. doi: 10.1016/j.vetmic.2004.08.014. Vet Microbiol. 2004. PMID: 15564023 Review.
-
Experimental animal infection models for Johne's disease, an infectious enteropathy caused by Mycobacterium avium subsp. paratuberculosis.Vet J. 2008 May;176(2):129-45. doi: 10.1016/j.tvjl.2007.02.022. Epub 2007 Apr 20. Vet J. 2008. PMID: 17449304 Review.
Cited by
-
Recombinant antibodies for diagnostics and therapy against pathogens and toxins generated by phage display.Proteomics Clin Appl. 2016 Oct;10(9-10):922-948. doi: 10.1002/prca.201600002. Epub 2016 Jun 21. Proteomics Clin Appl. 2016. PMID: 27198131 Free PMC article. Review.
-
Infectious disease antibodies for biomedical applications: A mini review of immune antibody phage library repertoire.Int J Biol Macromol. 2020 Nov 15;163:640-648. doi: 10.1016/j.ijbiomac.2020.06.268. Epub 2020 Jul 8. Int J Biol Macromol. 2020. PMID: 32650013 Free PMC article. Review.
-
Maximizing capture efficiency and specificity of magnetic separation for Mycobacterium avium subsp. paratuberculosis cells.Appl Environ Microbiol. 2010 Nov;76(22):7550-8. doi: 10.1128/AEM.01432-10. Epub 2010 Sep 17. Appl Environ Microbiol. 2010. PMID: 20851966 Free PMC article.
-
Developing Recombinant Antibodies by Phage Display Against Infectious Diseases and Toxins for Diagnostics and Therapy.Front Cell Infect Microbiol. 2021 Jul 7;11:697876. doi: 10.3389/fcimb.2021.697876. eCollection 2021. Front Cell Infect Microbiol. 2021. PMID: 34307196 Free PMC article. Review.
-
Neutralizing human recombinant antibodies against herpes simplex virus type 1 glycoproteins B from a phage-displayed scFv antibody library.Life Sci. 2017 Jan 15;169:1-5. doi: 10.1016/j.lfs.2016.11.018. Epub 2016 Nov 23. Life Sci. 2017. PMID: 27888111 Free PMC article.
References
-
- Barnes, P. F., and M. D. Cave. 2003. Molecular epidemiology of tuberculosis. N. Engl. J. Med. 349:1149-1156. - PubMed
-
- Cao, J., Y. Sun, T. Berglindh, B. Mellgard, Z. Li, B. Mardh, and S. Mardh. 2000. Helicobacter pylori-antigen-binding fragments expressed on the filamentous M13 phage prevent bacterial growth. Biochim. Biophys. Acta 1474:107-113. - PubMed
-
- Cummings, P. J., N. E. Hooper, and S. S. Rowland. 1998. Generation of a recombinant bacteriophage antibody library to Mycobacterium tuberculosis. Hybridoma 17:151-156. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

