Antipsychotic drugs disrupt normal development in Caenorhabditis elegans via additional mechanisms besides dopamine and serotonin receptors
- PMID: 16962336
- PMCID: PMC1694634
- DOI: 10.1016/j.phrs.2006.07.002
Antipsychotic drugs disrupt normal development in Caenorhabditis elegans via additional mechanisms besides dopamine and serotonin receptors
Abstract
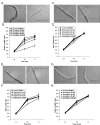
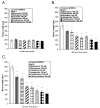
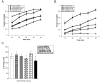
Antipsychotic drugs may produce adverse effects during development in humans and rodents. However, the extent of these effects has not been systematically characterized nor have molecular mechanisms been identified. Consequently, we sought to evaluate the effects of an extensive panel of antipsychotic drugs in a model organism, Caenorhabditis elegans, whose development is well characterized and which offers the possibility of identifying novel molecular targets. For these studies, animals were grown from hatching in the presence of vehicle (control) or antipsychotic drugs over a range of concentrations (20-160microM) and growth was analyzed by measuring head-to-tail length at various intervals. First-generation antipsychotics (e.g., fluphenazine) generally slowed growth and maturation more than second-generation drugs such as quetiapine and olanzapine. This is consistent with in vitro effects on human neuronal cell lines. Clozapine, a second-generation drug, produced similar growth deficits as haloperidol. Converging lines of evidence, including the failure to rescue growth with high concentrations of agonists, suggested that the drug-induced delay in development was not mediated by the major neurotransmitter receptors recognized by the antipsychotic drugs. Moreover, in serotonin-deficient tph-1 mutants, the drugs dramatically slowed development and led to larval arrest (including dauer formation) and neuronal abnormalities. Evaluation of alternative targets of the antipsychotics revealed a potential role for calmodulin and underscored the significance of Ca(2+)-calmodulin signaling in development. These findings suggest that antipsychotic drugs may interfere with normal developmental processes and provide a tool for investigating the key signaling pathways involved.
Conflict of interest statement
Conflict of interest statement: D.S.D. is funded by grants from NIH (HL84926 and MH68385) and Eli Lilly and Co.
Figures


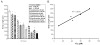
Similar articles
-
Antipsychotic drugs alter neuronal development including ALM neuroblast migration and PLM axonal outgrowth in Caenorhabditis elegans.Int J Dev Neurosci. 2008 May-Jun;26(3-4):371-80. doi: 10.1016/j.ijdevneu.2007.08.021. Epub 2008 Jan 20. Int J Dev Neurosci. 2008. PMID: 18282677 Free PMC article.
-
Antipsychotic drugs up-regulate tryptophan hydroxylase in ADF neurons of Caenorhabditis elegans: role of calcium-calmodulin-dependent protein kinase II and transient receptor potential vanilloid channel.J Neurosci Res. 2008 Aug 15;86(11):2553-63. doi: 10.1002/jnr.21684. J Neurosci Res. 2008. PMID: 18438926
-
Atypical antipsychotics: mechanism of action.Can J Psychiatry. 2002 Feb;47(1):27-38. Can J Psychiatry. 2002. PMID: 11873706 Review.
-
Social feeding in Caenorhabditis elegans is modulated by antipsychotic drugs and calmodulin and may serve as a protophenotype for asociality.Neuropharmacology. 2015 May;92:56-62. doi: 10.1016/j.neuropharm.2014.12.027. Epub 2015 Jan 7. Neuropharmacology. 2015. PMID: 25576370
-
Neuropharmacology of second-generation antipsychotic drugs: a validity of the serotonin-dopamine hypothesis.Prog Brain Res. 2008;172:199-212. doi: 10.1016/S0079-6123(08)00910-2. Prog Brain Res. 2008. PMID: 18772034 Review.
Cited by
-
Methods for studying the mechanisms of action of antipsychotic drugs in Caenorhabditis elegans.J Vis Exp. 2014 Feb 4;(84):e50864. doi: 10.3791/50864. J Vis Exp. 2014. PMID: 24561801 Free PMC article.
-
Drug elucidation: invertebrate genetics sheds new light on the molecular targets of CNS drugs.Front Pharmacol. 2014 Jul 28;5:177. doi: 10.3389/fphar.2014.00177. eCollection 2014. Front Pharmacol. 2014. PMID: 25120487 Free PMC article. Review.
-
Antipsychotic drugs alter neuronal development including ALM neuroblast migration and PLM axonal outgrowth in Caenorhabditis elegans.Int J Dev Neurosci. 2008 May-Jun;26(3-4):371-80. doi: 10.1016/j.ijdevneu.2007.08.021. Epub 2008 Jan 20. Int J Dev Neurosci. 2008. PMID: 18282677 Free PMC article.
-
Investigating the effects of antipsychotic drugs as a treatment for improving the activity of the unc-33 /Dpysl2 gene in C. elegans.MicroPubl Biol. 2024 Jul 31;2024:10.17912/micropub.biology.001063. doi: 10.17912/micropub.biology.001063. eCollection 2024. MicroPubl Biol. 2024. PMID: 39149410 Free PMC article.
-
Clozapine Modulates Glucosylceramide, Clears Aggregated Proteins, and Enhances ATG8/LC3 in Caenorhabditis elegans.Neuropsychopharmacology. 2017 Mar;42(4):951-962. doi: 10.1038/npp.2016.230. Epub 2016 Oct 6. Neuropsychopharmacology. 2017. PMID: 27711049 Free PMC article.
References
-
- Ansorge MS, Zhou M, Lira A, Hen R, Gingrich JA. Early-life blockade of the 5-HT transporter alters emotional behavior in adult mice. Science. 2004;306:879–881. - PubMed
-
- Committee on Drugs. Use of psychoactive medication during pregnancy and possible effects on the fetus and newborn. Pediatrics. 2000;105:880–887. - PubMed
-
- Kopelman AE, McCullar FW, Heggeness L. Limb malformations following maternal use of haloperidol. JAMA. 1975;231:62–63. - PubMed
-
- Elia J, Katz IR, Simpson GM. Teratogenicity of psychotherapeutic medications. Psychopharmacol Bull. 1987;23:531–586. - PubMed
-
- Godet PF, Marie-Cardine M. Neuroleptics, schizophrenia and pregnancy. Epidemiological and teratologic study. Encephale. 1991;17:543–547. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

