Aberrant contraction of antigen-specific CD4 T cells after infection in the absence of gamma interferon or its receptor
- PMID: 16966404
- PMCID: PMC1695510
- DOI: 10.1128/IAI.00847-06
Aberrant contraction of antigen-specific CD4 T cells after infection in the absence of gamma interferon or its receptor
Abstract
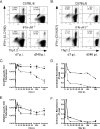
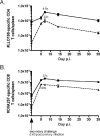
Several lines of evidence from different model systems suggest that gamma interferon (IFN-gamma) is an important regulator of T-cell contraction after antigen (Ag)-driven expansion. To specifically investigate the role of IFN-gamma in regulating the contraction of Ag-specific CD4 T cells, we infected IFN-gamma-/- and IFN-gammaR1-/- mice with attenuated Listeria monocytogenes and monitored the numbers of Ag-specific CD4 T cells during the expansion, contraction, and memory phases of the immune response to infection. In the absence of IFN-gamma or the ligand-binding portion of its receptor, Ag-specific CD4 T cells exhibited normal expansion in numbers, but in both strains of deficient mice there was very little decrease in the number of Ag-specific CD4 T cells even at time points later than day 90 after infection. This significant delay in contraction was not due to prolonged infection, since mice treated with antibiotics to conclusively eliminate infection exhibited the same defect in contraction. In addition to altering the number of Ag-specific CD4 T cells, the absence of IFN-gamma signaling also changed the phenotype of cells generated after infection. IFN-gammaR1-/- Ag-specific CD4 T cells reacquired expression of CD127 more quickly than wild-type cells, and more IFN-gammaR1-/- CD4 T cells were capable of producing both IFN-gamma and interleukin 2 following Ag stimulation. From these data we conclude that IFN-gamma regulates the contraction, phenotype, and function of Ag-specific CD4 T cells generated after infection.
Figures

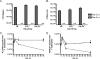



References
-
- Altman, J. D., P. A. Moss, P. J. Goulder, D. H. Barouch, M. G. McHeyzer-Williams, J. I. Bell, A. J. McMichael, and M. M. Davis. 1996. Phenotypic analysis of antigen-specific T lymphocytes. Science 274:94-96. - PubMed
-
- Bach, E. A., M. Aguet, and R. D. Schreiber. 1997. The IFN γ receptor: a paradigm for cytokine receptor signaling. Annu. Rev. Immunol. 15:563-591. - PubMed
-
- Bach, E. A., S. J. Szabo, A. S. Dighe, A. Ashkenazi, M. Aguet, K. M. Murphy, and R. D. Schreiber. 1995. Ligand-induced autoregulation of IFN-γ receptor β chain expression in T helper cell subsets. Science 270:1215-1218. - PubMed
-
- Badovinac, V. P., and J. T. Harty. 2000. Intracellular staining for TNF and IFN-γ detects different frequencies of antigen-specific CD8+ T cells. J. Immunol. Methods 238:107-117. - PubMed
-
- Badovinac, V. P., K. A. Messingham, S. E. Hamilton, and J. T. Harty. 2003. Regulation of CD8+ T cells undergoing primary and secondary responses to infection in the same host. J. Immunol. 170:4933-4942. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

