The C-terminus of murine S100A9 protein inhibits hyperalgesia induced by the agonist peptide of protease-activated receptor 2 (PAR2)
- PMID: 16967049
- PMCID: PMC1978433
- DOI: 10.1038/sj.bjp.0706884
The C-terminus of murine S100A9 protein inhibits hyperalgesia induced by the agonist peptide of protease-activated receptor 2 (PAR2)
Abstract
Background and purpose: S100A9 protein induces anti-nociception in rodents, in different experimental models of inflammatory pain. Herein, we investigated the effects of a fragment of the C-terminus of S100A9 (mS100A9p), on the hyperalgesia induced by serine proteases, through the activation of protease-activated receptor-2 (PAR2).
Experimental approach: Mechanical and thermal hyperalgesia induced by PAR2 agonists (SLIGRL-NH2 and trypsin) was measured in rats submitted to the paw pressure or plantar tests, and Egr-1 expression was determined by immunohistochemistry in rat spinal cord dorsal horn. Calcium flux in human embryonic kidney cells (HEK), which naturally express PAR2, in Kirsten virus-transformed kidney cells, transfected (KNRK-PAR2) or not (KNRK) with PAR2, and in mouse dorsal root ganglia neurons (DRG) was measured by fluorimetric methods.
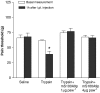
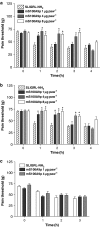
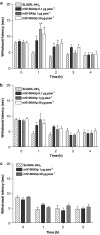
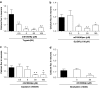
Key results: mS100A9p inhibited mechanical hyperalgesia induced by trypsin, without modifying its enzymatic activity. Mechanical and thermal hyperalgesia induced by SLIGRL-NH2 were inhibited by mS100A9p. SLIGRL-NH2 enhanced Egr-1 expression, a marker of nociceptor activation, and this effect was inhibited by concomitant treatment with mS100A9p. mS100A9p inhibited calcium mobilization in DRG neurons in response to the PAR2 agonists trypsin and SLIGRL-NH2, but also in response to capsaicin and bradykinin, suggesting a direct effect of mS100A9 on sensory neurons. No effect on the calcium flux induced by trypsin or SLIGRL in HEK cells or KNRK-PAR2 cells was observed.
Conclusions and implications: These data demonstrate that mS100A9p interferes with mechanisms involved in nociception and hyperalgesia and modulates, possibly directly on sensory neurons, the PAR2-induced nociceptive signal.
Figures



Similar articles
-
Protease-activated receptor 2 sensitizes the capsaicin receptor transient receptor potential vanilloid receptor 1 to induce hyperalgesia.J Neurosci. 2004 May 5;24(18):4300-12. doi: 10.1523/JNEUROSCI.5679-03.2004. J Neurosci. 2004. PMID: 15128844 Free PMC article.
-
Analgesic properties of S100A9 C-terminal domain: a mechanism dependent on calcium channel inhibition.Fundam Clin Pharmacol. 2009 Aug;23(4):427-38. doi: 10.1111/j.1472-8206.2009.00686.x. Epub 2009 Jun 8. Fundam Clin Pharmacol. 2009. PMID: 19515068
-
Effect of the C-terminus of murine S100A9 protein on experimental nociception.Peptides. 2006 Nov;27(11):2794-802. doi: 10.1016/j.peptides.2006.07.002. Epub 2006 Aug 21. Peptides. 2006. PMID: 16920226
-
The role of protease-activated receptor type 2 in nociceptive signaling and pain.Physiol Res. 2016 Jul 18;65(3):357-67. doi: 10.33549/physiolres.933269. Epub 2016 Apr 12. Physiol Res. 2016. PMID: 27070742 Review.
-
Protease-activated receptors and inflammatory hyperalgesia.Mem Inst Oswaldo Cruz. 2005 Mar;100 Suppl 1:173-6. doi: 10.1590/s0074-02762005000900029. Epub 2005 Jun 14. Mem Inst Oswaldo Cruz. 2005. PMID: 15962118 Review.
Cited by
-
Modulation of meningeal nociceptors mechanosensitivity by peripheral proteinase-activated receptor-2: the role of mast cells.Cephalalgia. 2008 Mar;28(3):276-84. doi: 10.1111/j.1468-2982.2007.01523.x. Cephalalgia. 2008. PMID: 18254896 Free PMC article.
-
Molecular Aspects Involved in the Mechanisms of Bothrops jararaca Venom-Induced Hyperalgesia: Participation of NK1 Receptor and Glial Cells.Toxins (Basel). 2024 Apr 10;16(4):187. doi: 10.3390/toxins16040187. Toxins (Basel). 2024. PMID: 38668612 Free PMC article.
-
The down regulation of neutrophil oxidative metabolism by S100A8 and S100A9: implication of the protease-activated receptor-2.Mol Immunol. 2012 Feb;50(1-2):42-8. doi: 10.1016/j.molimm.2011.12.001. Epub 2011 Dec 26. Mol Immunol. 2012. PMID: 22204866 Free PMC article.
-
Inhibition of macrophage functions by the C-terminus of murine S100A9 is dependent on B-1 cells.Mediators Inflamm. 2014;2014:836491. doi: 10.1155/2014/836491. Epub 2014 Sep 2. Mediators Inflamm. 2014. PMID: 25276056 Free PMC article.
References
-
- Aguiar-Passeti T, Postol E, Sorg C, Mariano M. Epithelioid cells from foreign-body granuloma selectively express the calcium-binding protein MRP-14, a novel down-regulatory molecule of macrophage activation. J Leukoc Biol. 1997;62:852–858. - PubMed
-
- Buritova J, Honore P, Besson JM. Indomethacin reduces both Krox-24 expression in the rat lumbar spinal cord and inflammatory signs following intraplantar carrageenan. Brain Res. 1995;674:211–220. - PubMed
-
- Brun JG, Haland G, Haga HJ, Fagerhol MK, Jonsson R. Effects of calprotectin in avridine-induced arthritis. APMIS. 1995;103:233–240. - PubMed
-
- Brun JG, Jonsson R, Haga HJ. Measurement of plasma calprotectin as an indicator of arthritis and disease activity in patients with inflammatory rheumatic diseases. J Rheumatol. 1994;21:733–738. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

