A therapeutic target for prostate cancer based on angiogenin-stimulated angiogenesis and cancer cell proliferation
- PMID: 16971483
- PMCID: PMC1599992
- DOI: 10.1073/pnas.0606708103
A therapeutic target for prostate cancer based on angiogenin-stimulated angiogenesis and cancer cell proliferation
Abstract
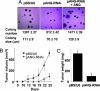
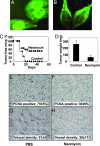
Human angiogenin is progressively up-regulated in the prostate epithelial cells during the development of prostate cancer from prostate intraepithelial neoplasia (PIN) to invasive adenocarcinoma. Mouse angiogenin is the most up-regulated gene in AKT-induced PIN in prostate-restricted AKT transgenic mice. These results prompted us to study the role that angiogenin plays in prostate cancer. Here, we report that, in addition to its well established role in mediating angiogenesis, angiogenin also directly stimulates prostate cancer cell proliferation. Angiogenin undergoes nuclear translocation in PC-3 human prostate cancer cells grown both in vitro and in mice. Thus, knocking down angiogenin expression in PC-3 human prostate adenocarcinoma cells inhibits ribosomal RNA transcription, in vitro cell proliferation, colony formation in soft agar, and xenograft growth in athymic mice. Blockade of nuclear translocation of angiogenin by the aminoglycoside antibiotic neomycin inhibited PC-3 cell tumor growth in athymic mice and was accompanied by a decrease in both cancer cell proliferation and angiogenesis. These results suggest that angiogenin has a dual effect, angiogenesis and cancer cell proliferation, in prostate cancer and may serve as a molecular target for drug development. Blocking nuclear translocation of angiogenin could have a combined benefit of antiangiogenesis and chemotherapy in treating prostate cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Fett JW, Strydom DJ, Lobb RR, Alderman EM, Bethune JL, Riordan JF, Vallee BL. Biochemistry. 1985;24:5480–5486. - PubMed
-
- Montero S, Guzman C, Cortes-Funes H, Colomer R. Clin Cancer Res. 1998;4:2161–2168. - PubMed
-
- Chopra V, Dinh TV, Hannigan EV. Cancer Invest. 1998;16:152–159. - PubMed
-
- Li D, Bell J, Brown A, Berry CL. J Pathol. 1994;172:171–175. - PubMed
-
- Shimoyama S, Shimizu N, Tsuji E, Yamasaki K, Kawahara M, Kaminishi M. Anticancer Res. 2002;22:1045–1052. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

