Context-dependent human extinction memory is mediated by a ventromedial prefrontal and hippocampal network
- PMID: 16971534
- PMCID: PMC2634865
- DOI: 10.1523/JNEUROSCI.2021-06.2006
Context-dependent human extinction memory is mediated by a ventromedial prefrontal and hippocampal network
Abstract
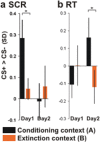
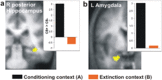
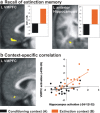
In fear extinction, an animal learns that a conditioned stimulus (CS) no longer predicts a noxious stimulus [unconditioned stimulus (UCS)] to which it had previously been associated, leading to inhibition of the conditioned response (CR). Extinction creates a new CS-noUCS memory trace, competing with the initial fear (CS-UCS) memory. Recall of extinction memory and, hence, CR inhibition at later CS encounters is facilitated by contextual stimuli present during extinction training. In line with theoretical predictions derived from animal studies, we show that, after extinction, a CS-evoked engagement of human ventromedial prefrontal cortex (VMPFC) and hippocampus is context dependent, being expressed in an extinction, but not a conditioning, context. Likewise, a positive correlation between VMPFC and hippocampal activity is extinction context dependent. Thus, a VMPFC-hippocampal network provides for context-dependent recall of human extinction memory, consistent with a view that hippocampus confers context dependence on VMPFC.
Figures

References
-
- Amaral DG. Introduction: what is where in the medial temporal lobe? Hippocampus. 1999;9:1–6. - PubMed
-
- Anagnostaras SG, Gale G, Fanselow MS. Hippocampus and contextual fear conditioning: recent controversies and advances. Hippocampus. 2001;11:8–17. - PubMed
-
- Ashburner J, Friston KJ, Penny W. Imaging neuroscience—theory and analysis. In: Frackowiak RS, Friston KJ, Frith C, Dolan RJ, Price CJ, editors. Human brain function. San Diego: Academic; 2004. pp. 599–1104.
-
- Boshuisen ML, Ter Horst GJ, Paans AM, Reinders AA, Den Boer JA. rCBF differences between panic disorder patients and control subjects during anticipatory anxiety and rest. Biol Psychiatry. 2002;52:126–135. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
