GroES/GroEL and DnaK/DnaJ have distinct roles in stress responses and during cell cycle progression in Caulobacter crescentus
- PMID: 16980445
- PMCID: PMC1698207
- DOI: 10.1128/JB.00824-06
GroES/GroEL and DnaK/DnaJ have distinct roles in stress responses and during cell cycle progression in Caulobacter crescentus
Abstract
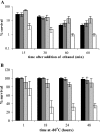
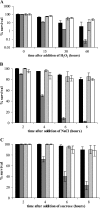
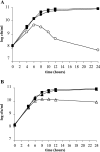
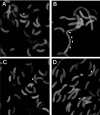
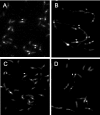
Misfolding and aggregation of protein molecules are major threats to all living organisms. Therefore, cells have evolved quality control systems for proteins consisting of molecular chaperones and proteases, which prevent protein aggregation by either refolding or degrading misfolded proteins. DnaK/DnaJ and GroES/GroEL are the best-characterized molecular chaperone systems in bacteria. In Caulobacter crescentus these chaperone machines are the products of essential genes, which are both induced by heat shock and cell cycle regulated. In this work, we characterized the viabilities of conditional dnaKJ and groESL mutants under different types of environmental stress, as well as under normal physiological conditions. We observed that C. crescentus cells with GroES/EL depleted are quite resistant to heat shock, ethanol, and freezing but are sensitive to oxidative, saline, and osmotic stresses. In contrast, cells with DnaK/J depleted are not affected by the presence of high concentrations of hydrogen peroxide, NaCl, and sucrose but have a lower survival rate after heat shock, exposure to ethanol, and freezing and are unable to acquire thermotolerance. Cells lacking these chaperones also have morphological defects under normal growth conditions. The absence of GroE proteins results in long, pinched filamentous cells with several Z-rings, whereas cells lacking DnaK/J are only somewhat more elongated than normal predivisional cells, and most of them do not have Z-rings. These findings indicate that there is cell division arrest, which occurs at different stages depending on the chaperone machine affected. Thus, the two chaperone systems have distinct roles in stress responses and during cell cycle progression in C. crescentus.
Figures

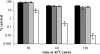


Similar articles
-
Complementation studies of the DnaK-DnaJ-GrpE chaperone machineries from Vibrio harveyi and Escherichia coli, both in vivo and in vitro.Arch Microbiol. 2004 Dec;182(6):436-49. doi: 10.1007/s00203-004-0727-8. Epub 2004 Sep 24. Arch Microbiol. 2004. PMID: 15448982
-
Defense against protein carbonylation by DnaK/DnaJ and proteases of the heat shock regulon.J Bacteriol. 2005 Jun;187(12):4207-13. doi: 10.1128/JB.187.12.4207-4213.2005. J Bacteriol. 2005. PMID: 15937182 Free PMC article.
-
Cells lacking ClpB display a prolonged shutoff phase of the heat shock response in Caulobacter crescentus.Mol Microbiol. 2005 Jul;57(2):592-603. doi: 10.1111/j.1365-2958.2005.04713.x. Mol Microbiol. 2005. PMID: 15978087
-
Review: mechanisms of disaggregation and refolding of stable protein aggregates by molecular chaperones.J Struct Biol. 2001 Aug;135(2):84-93. doi: 10.1006/jsbi.2001.4352. J Struct Biol. 2001. PMID: 11580258 Review.
-
Review: a structural view of the GroE chaperone cycle.J Struct Biol. 2001 Aug;135(2):95-103. doi: 10.1006/jsbi.2001.4387. J Struct Biol. 2001. PMID: 11580259 Review.
Cited by
-
De Novo Structural Pattern Mining in Cellular Electron Cryotomograms.Structure. 2019 Apr 2;27(4):679-691.e14. doi: 10.1016/j.str.2019.01.005. Epub 2019 Feb 7. Structure. 2019. PMID: 30744995 Free PMC article.
-
Age-Dependent Pleomorphism in Mycobacterium monacense Cultures.Microorganisms. 2025 Feb 20;13(3):475. doi: 10.3390/microorganisms13030475. Microorganisms. 2025. PMID: 40142368 Free PMC article.
-
Genomic Signatures of Adaptation to Stress Reveal Shared Evolutionary Trends Between Tetrahymena utriculariae and Its Algal Endosymbiont, Micractinium tetrahymenae.Mol Biol Evol. 2025 Feb 3;42(2):msaf030. doi: 10.1093/molbev/msaf030. Mol Biol Evol. 2025. PMID: 39895309 Free PMC article.
-
Growth of sulfate-reducing Desulfobacterota and Bacillota at periodic oxygen stress of 50% air-O2 saturation.Microbiome. 2024 Oct 4;12(1):191. doi: 10.1186/s40168-024-01909-7. Microbiome. 2024. PMID: 39367500 Free PMC article.
-
Ancestors in the Extreme: A Genomics View of Microbial Diversity in Hypersaline Aquatic Environments.Results Probl Cell Differ. 2024;71:185-212. doi: 10.1007/978-3-031-37936-9_10. Results Probl Cell Differ. 2024. PMID: 37996679
References
-
- Avedissian, M., and S. L. Gomes. 1996. Expression of the groESL operon is cell-cycle controlled in Caulobacter crescentus. Mol. Microbiol. 19:79-89. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

