Therapeutic intervention based on protein prenylation and associated modifications
- PMID: 16983387
- PMCID: PMC2892741
- DOI: 10.1038/nchembio818
Therapeutic intervention based on protein prenylation and associated modifications
Abstract
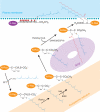
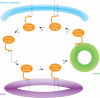
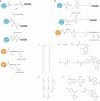
In eukaryotic cells, a specific set of proteins are modified by C-terminal attachment of 15-carbon farnesyl groups or 20-carbon geranylgeranyl groups that function both as anchors for fixing proteins to membranes and as molecular handles for facilitating binding of these lipidated proteins to other proteins. Additional modification of these prenylated proteins includes C-terminal proteolysis and methylation, and attachment of a 16-carbon palmitoyl group; these modifications augment membrane anchoring and alter the dynamics of movement of proteins between different cellular membrane compartments. The enzymes in the protein prenylation pathway have been isolated and characterized. Blocking protein prenylation is proving to be therapeutically useful for the treatment of certain cancers, infection by protozoan parasites and the rare genetic disease Hutchinson-Gilford progeria syndrome.
Figures


References
-
- Sakagami Y, et al. Isolation of a novel sex hormone tremerogen A-10, controlling conjugation tube formation in Tremella mesenterica fries. Agric. Biol. Chem. 1978;42:1093–1094.
-
- Tsuchiya E, Fukui S, Kamiya Y, Sakagami Y, Fujino M. Requirements of chemical structure for hormonal activity of lipo-peptidyl factors inducing sexual differentiation in vegetative cells of heterobasidiomycetous yeasts. Biochem. Biophys. Res. Commun. 1978;85:459–463. - PubMed
-
- Anderegg RJ, Betz R, Carr SA, Crabb JW, Duntze W. Structure of Saccharomyces cerevisiae mating hormone a-factor. Identification of S-farnesyl cysteine as a structural component. J. Biol. Chem. 1988;263:18236–18240. - PubMed
-
- Glomset JA, Gelb MH, Farnsworth CC. Prenyl proteins in eukaryotic cells: a new type of membrane anchor. Trends Biochem. Sci. 1990;15:139–142. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

