The use of an acellular dermal regenerative tissue matrix in the treatment of lower extremity wounds: a prospective 16-week pilot study
- PMID: 16984575
- PMCID: PMC7951702
- DOI: 10.1111/j.1742-481X.2006.00209.x
The use of an acellular dermal regenerative tissue matrix in the treatment of lower extremity wounds: a prospective 16-week pilot study
Abstract
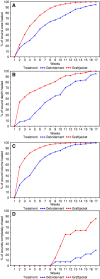
A prospective, single-centre, randomized controlled study was performed to evaluate the effectiveness of Graftjacket, a human acellular regenerative tissue matrix as a treatment option for chronic non healing lower extremity wounds. Twenty-eight diabetic patients with full-thickness wounds that had been present for at least 6 weeks were treated with sharp debridement and randomized to a single application of Graftjacket tissue matrix plus mineral oil-soaked fluff compression dressing or to a control treatment of wound gel with gauze dressings. All patients were seen weekly. By week 16, 12 of 14 patients treated with Graftjacket tissue matrix demonstrated complete wound closure compared with 4 of 14 patients in the control group. Patients treated with Graftjacket tissue matrix showed a statistically significant higher percentage of wound healing with respect to wound area, and clinically significant differences in wound depth and wound volume. This comparison is not performed to demonstrate that the application of the Grafjacket is more effective than sharp debridement. This study is done to help assign a role to the use of Graftjacket matrix in lower extremity wound care.
Figures
References
-
- Sanders LJ, Reiber GE. Diabetic Foot Ulcers and Amputations. American Diabetes Association, Inc, 2001.
-
- Gentzkow GD, Iwasaki SD, Hershon KS, Mengel M, Prendergast JJ, Ricotta JJ, Steed DP, Lipkin S. Use of dermagraft, a cultured human dermis, to treat diabetic foot ulcers. Diabetes Care 1996;19:350–4. - PubMed
-
- Marston WA, Hanft J, Norwood P, Pollak R. The efficacy and safety of Dermagraft in improving the healing of chronic diabetic foot ulcers: results of a prospective randomized trial. Diabetes Care 2003;26:1701–5. - PubMed
-
- Grey JE, Lowe G, Bale S, Harding KG. The use of cultured dermis in the treatment of diabetic foot ulcers. J Wound Care 1998;7:324–5. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

