Identification of sequence motifs that target neuronal nicotinic receptors to dendrites and axons
- PMID: 16988049
- PMCID: PMC6674458
- DOI: 10.1523/JNEUROSCI.0840-06.2006
Identification of sequence motifs that target neuronal nicotinic receptors to dendrites and axons
Abstract
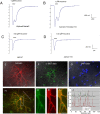
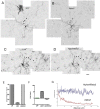
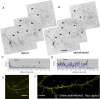
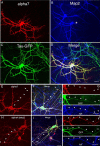
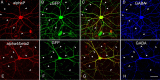
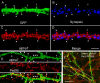
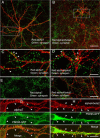
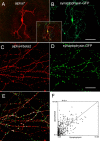
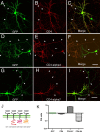
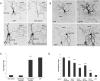
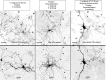
Neuronal nicotinic acetylcholine receptors (nAChRs) belong to a family of ligand-gated ion channels that play important roles in central and peripheral nervous systems. The subcellular distribution of neuronal nAChRs has important implications for function and is not well understood. Here, we analyzed the targeting of two major types of neuronal nAChRs by expressing epitope-tagged subunits in cultured hippocampal neurons. Surprisingly, the alpha7 nAChR (alpha7) and alpha4/beta2 nAChR (alpha4beta2) displayed distinct patterns of expression, with alpha7 targeted preferentially to the somatodendritic compartments, whereas alpha4beta2 was localized to both axonal and dendritic domains. When fused to CD4 or IL2RA (interleukin 2 receptor alpha subunit) proteins, which are normally distributed ubiquitously, the M3-M4 intracellular loop from the alpha7 subunit promoted dendritic expression, whereas the homologous M3-M4 loop from the alpha4 subunit led to surface axonal expression. Systemic screening and alanine substitution further identified a 25-residue leucine motif ([DE]XXXL[LI]) containing an axonal targeting sequence within the alpha4 loop and a 48-residue dileucine and tyrosine motif (YXXØ) containing a dendritic targeting sequence from the alpha7 loop. These results provide valuable information in understanding diverse roles of neuronal nAChRs in mediating and modulating synaptic transmission, synaptic plasticity, and nicotine addiction.
Figures


References
-
- Alkondon M, Albuquerque EX. Diversity of nicotinic acetylcholine receptors in rat hippocampal neurons. I. Pharmacological and functional evidence for distinct structural subtypes. J Pharmacol Exp Ther. 1993;265:1455–1473. - PubMed
-
- Alkondon M, Pereira EF, Barbosa CT, Albuquerque EX. Neuronal nicotinic acetylcholine receptor activation modulates gamma-aminobutyric acid release from CA1 neurons of rat hippocampal slices. J Pharmacol Exp Ther. 1997;283:1396–1411. - PubMed
-
- Alkondon M, Pereira EF, Almeida LE, Randall WR, Albuquerque EX. Nicotine at concentrations found in cigarette smokers activates and desensitizes nicotinic acetylcholine receptors in CA1 interneurons of rat hippocampus. Neuropharmacology. 2000;39:2726–2739. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
