Measurement of effector protein injection by type III and type IV secretion systems by using a 13-residue phosphorylatable glycogen synthase kinase tag
- PMID: 16988240
- PMCID: PMC1594927
- DOI: 10.1128/IAI.00690-06
Measurement of effector protein injection by type III and type IV secretion systems by using a 13-residue phosphorylatable glycogen synthase kinase tag
Abstract
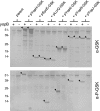
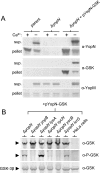
Numerous bacterial pathogens use type III secretion systems (T3SSs) or T4SSs to inject or translocate virulence proteins into eukaryotic cells. Several different reporter systems have been developed to measure the translocation of these proteins. In this study, a peptide tag-based reporter system was developed and used to monitor the injection of T3S and T4S substrates. The glycogen synthase kinase (GSK) tag is a 13-residue phosphorylatable peptide tag derived from the human GSK-3beta kinase. Translocation of a GSK-tagged protein into a eukaryotic cell results in host cell protein kinase-dependent phosphorylation of the tag, which can be detected with phosphospecific GSK-3beta antibodies. A series of expression plasmids encoding Yop-GSK fusion proteins were constructed to evaluate the ability of the GSK tag to measure the injection of Yops by the Yersinia pestis T3SS. GSK-tagged YopE, YopH, LcrQ, YopK, YopN, and YopJ were efficiently phosphorylated when translocated into HeLa cells. Similarly, the injection of GSK-CagA by the Helicobacter pylori T4SS into different cell types was measured via phosphorylation of the GSK tag. The GSK tag provides a simple method to monitor the translocation of T3S and T4S substrates.
Figures





References
-
- Akeda, Y., and J. E. Galan. 2005. Chaperone release and unfolding of substrates in type III secretion. Nature 437:911-915. - PubMed
-
- Ali, S. A., and A. Steinkasserer. 1995. PCR-ligation-PCR mutagenesis: a protocol for creating gene fusions and mutations. BioTechniques 18:746-750. - PubMed
-
- Boyd, A. P., N. Grosdent, S. Totemeyer, C. Geuijen, S. Bleves, M. Iriarte, I. Lambermont, J. N. Octave, and G. R. Cornelis. 2000. Yersinia enterocolitica can deliver Yop proteins into a wide range of cell types: development of a delivery system for heterologous proteins. Eur. J. Cell Biol. 79:659-671. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

