Activated human T lymphocytes express cyclooxygenase-2 and produce proadipogenic prostaglandins that drive human orbital fibroblast differentiation to adipocytes
- PMID: 17003477
- PMCID: PMC1698858
- DOI: 10.2353/ajpath.2006.060434
Activated human T lymphocytes express cyclooxygenase-2 and produce proadipogenic prostaglandins that drive human orbital fibroblast differentiation to adipocytes
Abstract
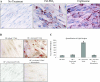
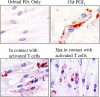
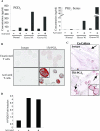
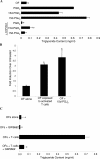
The differentiation of preadipocyte fibroblasts to adipocytes is a crucial process to many disease states including obesity, cardiovascular, and autoimmune diseases. In Graves' disease, the orbit of the eye can become severely inflamed and infiltrated with T lymphocytes as part of the autoimmune process. The orbital fibroblasts convert to fat-like cells causing the eye to protrude, which is disfiguring and can lead to blindness. Recently, the transcription factor peroxisome proliferator activated receptor (PPAR)-gamma and its natural (15d-PGJ2) and synthetic (thiazolidinedione-type) PPAR-gamma agonists have been shown to be crucial to the in vitro differentiation of preadipocyte fibroblasts to adipocytes. We show herein several novel findings. First, that activated T lymphocytes from Graves' patients drive the differentiation of PPAR-gamma-expressing orbital fibroblasts to adipocytes. Second, this adipogenic differentiation is blocked by nonselective small molecule cyclooxygenase (Cox)-1/Cox-2 inhibitors and by Cox-2 selective inhibitors. Third, activated, but not naïve, human T cells highly express Cox-2 and synthesize prostaglandin D2 and related prostaglandins that are PPAR-gamma ligands. These provocative new findings provide evidence for how activated T lymphocytes, through production of PPAR-gamma ligands, profoundly influence human fibroblast differentiation to adipocytes. They also suggest the possibility that, in addition to the orbit, T lymphocytes influence the deposition of fat in other tissues.
Figures




References
-
- Gabbiani G. The myofibroblast: a key cell for wound healing and fibrocontractive diseases. Prog Clin Biol Res. 1981;54:183–194. - PubMed
-
- Phan SH. The myofibroblast in pulmonary fibrosis. Chest. 2002;122:286S–289S. - PubMed
-
- Brody JS, Kaplan NB. Proliferation of alveolar interstitial cells during postnatal lung growth. Evidence for two distinct populations of pulmonary fibroblasts. Am Rev Respir Dis. 1983;127:763–770. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

