Respiratory syncytial virus F envelope protein associates with lipid rafts without a requirement for other virus proteins
- PMID: 17005642
- PMCID: PMC1676292
- DOI: 10.1128/JVI.00643-06
Respiratory syncytial virus F envelope protein associates with lipid rafts without a requirement for other virus proteins
Abstract
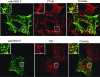
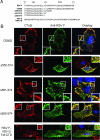
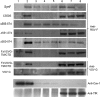
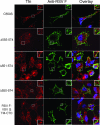
Like many enveloped viruses, human respiratory syncytial virus (RSV) assembles at and buds from lipid rafts. Translocation of the envelope proteins to these membrane subdomains is essential for production of infectious virus, but the targeting mechanism is poorly understood and it is not known if other virus proteins are required. Here we demonstrate that F protein of RSV intrinsically targets to lipid rafts without a requirement for any other virus protein, including the SH and G envelope proteins. Recombinant virus deficient in SH and G but retaining F protein expression was used to demonstrate that F protein still localized in rafts in both A549 and HEp-2 cells. Expression of a recombinant F gene by use of plasmid vectors demonstrated that F contains its own targeting domain and localized to rafts in the absence of other virus proteins. The domain responsible for translocation was then mapped. Unlike most other virus envelope proteins, F is unusual since the target signal is not contained within the cytoplasmic domain nor did it involve fatty acid modified residues. Furthermore, exchange of the transmembrane domain with that of the vesicular stomatitis virus G protein, a nonraft protein, did not alter F protein raft localization. Taken together, these data suggest that domains present in the extracellular portion of the protein are responsible for lipid raft targeting of the RSV F protein.
Figures


Similar articles
-
The cytoplasmic tail of the human respiratory syncytial virus F protein plays critical roles in cellular localization of the F protein and infectious progeny production.J Virol. 2006 Nov;80(21):10465-77. doi: 10.1128/JVI.01439-06. Epub 2006 Aug 23. J Virol. 2006. PMID: 16928754 Free PMC article.
-
Influenza virus hemagglutinin concentrates in lipid raft microdomains for efficient viral fusion.Proc Natl Acad Sci U S A. 2003 Dec 9;100(25):14610-7. doi: 10.1073/pnas.2235620100. Epub 2003 Oct 15. Proc Natl Acad Sci U S A. 2003. PMID: 14561897 Free PMC article.
-
Decoding respiratory syncytial virus morphology: distinct structural and molecular signatures of spherical and filamentous particles.Front Cell Infect Microbiol. 2025 May 27;15:1597279. doi: 10.3389/fcimb.2025.1597279. eCollection 2025. Front Cell Infect Microbiol. 2025. PMID: 40496018 Free PMC article.
-
Secretion of the respiratory syncytial virus fusion protein from insect cells using the baculovirus expression system.Methods Mol Biol. 2007;379:149-61. doi: 10.1007/978-1-59745-393-6_11. Methods Mol Biol. 2007. PMID: 17502677 Review.
-
[Research Progress in the F Gene and Protein of the Respiratory Syncytial Virus].Bing Du Xue Bao. 2015 Mar;31(2):201-6. Bing Du Xue Bao. 2015. PMID: 26164949 Review. Chinese.
Cited by
-
Canine distemper virus infection requires cholesterol in the viral envelope.J Virol. 2007 Apr;81(8):4158-65. doi: 10.1128/JVI.02647-06. Epub 2007 Jan 31. J Virol. 2007. PMID: 17267508 Free PMC article.
-
All Eyes on the Prefusion-Stabilized F Construct, but Are We Missing the Potential of Alternative Targets for Respiratory Syncytial Virus Vaccine Design?Vaccines (Basel). 2024 Jan 18;12(1):97. doi: 10.3390/vaccines12010097. Vaccines (Basel). 2024. PMID: 38250910 Free PMC article. Review.
-
A role for caveolin 1 in assembly and budding of the paramyxovirus parainfluenza virus 5.J Virol. 2010 Oct;84(19):9749-59. doi: 10.1128/JVI.01079-10. Epub 2010 Jul 14. J Virol. 2010. PMID: 20631121 Free PMC article.
-
Statin-mediated disruption of Rho GTPase prenylation and activity inhibits respiratory syncytial virus infection.Commun Biol. 2021 Oct 29;4(1):1239. doi: 10.1038/s42003-021-02754-2. Commun Biol. 2021. PMID: 34716403 Free PMC article.
-
Cholesterol is required for stability and infectivity of influenza A and respiratory syncytial viruses.Virology. 2017 Oct;510:234-241. doi: 10.1016/j.virol.2017.07.024. Epub 2017 Jul 25. Virology. 2017. PMID: 28750327 Free PMC article.
References
-
- Alexander, M., Y. C. Bor, K. S. Ravichandran, M. L. Hammarskjold, and D. Rekosh. 2004. Human immunodeficiency virus type 1 Nef associates with lipid rafts to downmodulate cell surface CD4 and class I major histocompatibility complex expression and to increase viral infectivity. J. Virol. 78:1685-1696. - PMC - PubMed
-
- Arumugham, R. G., R. C. J. Seid, S. Doyle, S. W. Hildreth, and P. R. Paradiso. 1989. Fatty acid acylation of the fusion glycoprotein of human respiratory syncytial virus. J. Biol. Chem. 264:10339-10342. - PubMed
-
- Brown, D. A., and E. London. 1998. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14:111-136. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

