Casein kinase 2 (CK2) increases survivin expression via enhanced beta-catenin-T cell factor/lymphoid enhancer binding factor-dependent transcription
- PMID: 17005722
- PMCID: PMC1622780
- DOI: 10.1073/pnas.0606845103
Casein kinase 2 (CK2) increases survivin expression via enhanced beta-catenin-T cell factor/lymphoid enhancer binding factor-dependent transcription
Abstract
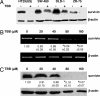
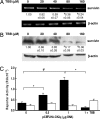
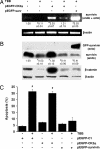
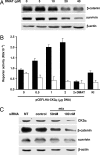
Increased expression of casein kinase 2 (CK2) is associated with hyperproliferation and suppression of apoptosis in cancer. Mutations in the tumor suppressor APC (adenomatous polyposis coli) are frequent in colon cancer and often augment beta-catenin-T cell factor (Tcf)/lymphoid enhancer binding factor (Lef)-dependent transcription of genes such as c-myc and cyclin-D1. CK2 has also been implicated recently in the regulation of beta-catenin stability. To identify mechanisms by which CK2 promotes survival, effects of the specific CK2 inhibitors 4,5,6,7-tetrabromobenzotriazole (TBB) and 2-dimethylamino-4,5,6,7-tetrabromo-1H-benzimidazole were assessed. TBB and 2-dimethylamino-4,5,6,7-tetrabromo-1H-benzimidazole significantly decreased proliferation and increased apoptosis of HT29(US) colon cancer cells. RT-PCR and immunoblot analysis revealed that both inhibitors decreased survivin mRNA and protein levels in HT29(US) cells. Similar effects were observed with TBB in human DLD-1 and SW-480 colorectal cells as well as ZR-75 breast cancer cells and HEK-293T embryonic kidney cells. Expression of GFP-CK2alpha in HEK-293T cells resulted in beta-catenin-Tcf/Lef-dependent up-regulation of survivin and increased resistance to anticancer drugs. Augmented beta-catenin-Tcf/Lef-dependent transcription and resistance to apoptosis observed upon GFP-CK2alpha expression were abolished by TBB. Alternatively, HEK-293T cells expressing GFP-survivin were resistant to TBB-induced apoptosis. Finally, siRNA-mediated down-regulation of CK2alpha in HEK-293T cells coincided with reduced beta-catenin and survivin levels. Taken together, these results suggest that CK2 kinase activity promotes survival by increasing survivin expression via beta-catenin-Tcf/Lef-mediated transcription. Hence, selective CK2 inhibition or down-regulation in tumors may provide an attractive opportunity for the development of novel cancer therapies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Caveolin-1-mediated suppression of cyclooxygenase-2 via a beta-catenin-Tcf/Lef-dependent transcriptional mechanism reduced prostaglandin E2 production and survivin expression.Mol Biol Cell. 2009 Apr;20(8):2297-310. doi: 10.1091/mbc.e08-09-0939. Epub 2009 Feb 25. Mol Biol Cell. 2009. PMID: 19244345 Free PMC article.
-
E-cadherin is required for caveolin-1-mediated down-regulation of the inhibitor of apoptosis protein survivin via reduced beta-catenin-Tcf/Lef-dependent transcription.Mol Cell Biol. 2007 Nov;27(21):7703-17. doi: 10.1128/MCB.01991-06. Epub 2007 Sep 4. Mol Cell Biol. 2007. PMID: 17785436 Free PMC article.
-
Protein kinase CK2 promotes cancer cell viability via up-regulation of cyclooxygenase-2 expression and enhanced prostaglandin E2 production.J Cell Biochem. 2011 Nov;112(11):3167-75. doi: 10.1002/jcb.23247. J Cell Biochem. 2011. PMID: 21732411
-
Calcium, calcium-sensing receptor and colon cancer.Cancer Lett. 2009 Mar 8;275(1):9-16. doi: 10.1016/j.canlet.2008.07.001. Epub 2008 Aug 23. Cancer Lett. 2009. PMID: 18725175 Review.
-
Diversity of LEF/TCF action in development and disease.Oncogene. 2006 Dec 4;25(57):7492-504. doi: 10.1038/sj.onc.1210056. Oncogene. 2006. PMID: 17143293 Review.
Cited by
-
Evaluation of protein kinase CK2 as a therapeutic target for squamous cell carcinoma of cats.Am J Vet Res. 2017 Aug;78(8):946-953. doi: 10.2460/ajvr.78.8.946. Am J Vet Res. 2017. PMID: 28738012 Free PMC article.
-
Aging brain microenvironment decreases hippocampal neurogenesis through Wnt-mediated survivin signaling.Aging Cell. 2012 Jun;11(3):542-52. doi: 10.1111/j.1474-9726.2012.00816.x. Epub 2012 Apr 4. Aging Cell. 2012. PMID: 22404871 Free PMC article.
-
Licoricidin improves neurological dysfunction after traumatic brain injury in mice via regulating FoxO3/Wnt/β-catenin pathway.J Nat Med. 2020 Sep;74(4):767-776. doi: 10.1007/s11418-020-01434-5. Epub 2020 Jul 12. J Nat Med. 2020. PMID: 32656716
-
ARF6-regulated endocytosis of growth factor receptors links cadherin-based adhesion to canonical Wnt signaling in epithelia.Mol Cell Biol. 2013 Aug;33(15):2963-75. doi: 10.1128/MCB.01698-12. Epub 2013 May 28. Mol Cell Biol. 2013. PMID: 23716594 Free PMC article.
-
Caveolin-1-mediated suppression of cyclooxygenase-2 via a beta-catenin-Tcf/Lef-dependent transcriptional mechanism reduced prostaglandin E2 production and survivin expression.Mol Biol Cell. 2009 Apr;20(8):2297-310. doi: 10.1091/mbc.e08-09-0939. Epub 2009 Feb 25. Mol Biol Cell. 2009. PMID: 19244345 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

