GABAA receptor alpha 4 subunits mediate extrasynaptic inhibition in thalamus and dentate gyrus and the action of gaboxadol
- PMID: 17005728
- PMCID: PMC1578762
- DOI: 10.1073/pnas.0604304103
GABAA receptor alpha 4 subunits mediate extrasynaptic inhibition in thalamus and dentate gyrus and the action of gaboxadol
Abstract
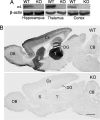
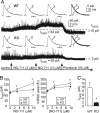
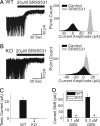
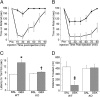
The neurotransmitter GABA mediates the majority of rapid inhibition in the CNS. Inhibition can occur via the conventional mechanism, the transient activation of subsynaptic GABAA receptors (GABAA-Rs), or via continuous activation of high-affinity receptors by low concentrations of ambient GABA, leading to "tonic" inhibition that can control levels of excitability and network activity. The GABAA-R alpha4 subunit is expressed at high levels in the dentate gyrus and thalamus and is suspected to contribute to extrasynaptic GABAA-R-mediated tonic inhibition. Mice were engineered to lack the alpha4 subunit by targeted disruption of the Gabra4 gene. alpha4 Subunit knockout mice are viable, breed normally, and are superficially indistinguishable from WT mice. In electrophysiological recordings, these mice show a lack of tonic inhibition in dentate granule cells and thalamic relay neurons. Behaviorally, knockout mice are insensitive to the ataxic, sedative, and analgesic effects of the novel hypnotic drug, gaboxadol. These data demonstrate that tonic inhibition in dentate granule cells and thalamic relay neurons is mediated by extrasynaptic GABAA-Rs containing the alpha4 subunit and that gaboxadol achieves its effects via the activation of this GABAA-R subtype.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

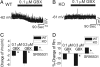
References
Publication types
MeSH terms
Substances
Grants and funding
- K08 DE014184/DE/NIDCR NIH HHS/United States
- I01 BX000404/BX/BLRD VA/United States
- P01 NS035985/NS/NINDS NIH HHS/United States
- R01 AA013004/AA/NIAAA NIH HHS/United States
- R37 AA007680/AA/NIAAA NIH HHS/United States
- R01 AA007680/AA/NIAAA NIH HHS/United States
- AA13004/AA/NIAAA NIH HHS/United States
- NS35985/NS/NINDS NIH HHS/United States
- R01 GM045129/GM/NIGMS NIH HHS/United States
- DE14184/DE/NIDCR NIH HHS/United States
- AA07680/AA/NIAAA NIH HHS/United States
- GM45129/GM/NIGMS NIH HHS/United States
- R01 AA013646/AA/NIAAA NIH HHS/United States
- AA13646/AA/NIAAA NIH HHS/United States
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

