Alpha-internexin is structurally and functionally associated with the neurofilament triplet proteins in the mature CNS
- PMID: 17005864
- PMCID: PMC6674481
- DOI: 10.1523/JNEUROSCI.2580-06.2006
Alpha-internexin is structurally and functionally associated with the neurofilament triplet proteins in the mature CNS
Abstract
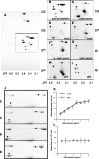
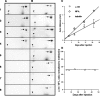
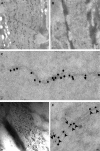
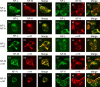
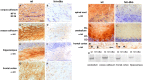
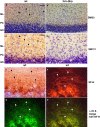
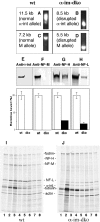
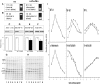
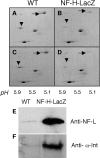
Alpha-internexin, a neuronal intermediate filament protein implicated in neurodegenerative disease, coexists with the neurofilament (NF) triplet proteins (NF-L, NF-M, and NF-H) but has an unknown function. The earlier peak expression of alpha-internexin than the triplet during brain development and its ability to form homopolymers, unlike the triplet, which are obligate heteropolymers, have supported a widely held view that alpha-internexin and neurofilament triplet form separate filament systems. Here, we demonstrate, however, that despite a postnatal decline in expression, alpha-internexin is as abundant as the triplet in the adult CNS and exists in a relatively fixed stoichiometry with these subunits. Alpha-internexin exhibits transport and turnover rates identical to those of triplet proteins in optic axons and colocalizes with NF-M on single neurofilaments by immunogold electron microscopy. Alpha-internexin also coassembles with all three neurofilament proteins into a single network of filaments in quadruple-transfected SW13vim(-) cells. Genetically deleting NF-M alone or together with NF-H in mice dramatically reduces alpha-internexin transport and content in axons throughout the CNS. Moreover, deleting alpha-internexin potentiates the effects of NF-M deletion on NF-H and NF-L transport. Finally, overexpressing a NF-H-LacZ fusion protein in mice induces alpha-internexin and neurofilament triplet to aggregate in neuronal perikarya and greatly reduces their transport and content selectively in axons. Our data show that alpha-internexin and the neurofilament proteins are functionally interdependent. The results strongly support the view that alpha-internexin is a fourth subunit of neurofilaments in the adult CNS, providing a basis for its close relationship with neurofilaments in CNS diseases associated with neurofilament accumulation.
Figures


References
-
- Al-Chalabi A, Miller CC. Neurofilaments and neurological disease. BioEssays. 2003;25:346–355. - PubMed
-
- Archer DR, Watson DF, Griffin JW. Phosphorylation-dependent immunoreactivity of neurofilaments and the rate of slow axonal transport in the central and peripheral axons of the rat dorsal root ganglion. J Neurochem. 1994;62:1119–1125. - PubMed
-
- Beaulieu JM, Robertson J, Julien JP. Interactions between peripherin and neurofilaments in cultured cells: disruption of peripherin assembly by the NF-M and NF-H subunits. Biochem Cell Biol. 1999;77:41–45. - PubMed
-
- Benson DL, Mandell JW, Shaw G, Banker G. Compartmentation of alpha-internexin and neurofilament triplet proteins in cultured hippocampal neurons. J Neurocytol. 1996;25:181–196. - PubMed
-
- Brown A. Visualization of single neurofilaments by immunofluorescence microscopy of splayed axonal cytoskeletons. Cell Motil Cytoskeleton. 1997;38:133–145. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
