Modelling spinal circuitry involved in locomotor pattern generation: insights from the effects of afferent stimulation
- PMID: 17008375
- PMCID: PMC1890432
- DOI: 10.1113/jphysiol.2006.118711
Modelling spinal circuitry involved in locomotor pattern generation: insights from the effects of afferent stimulation
Abstract
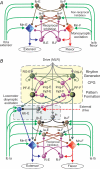
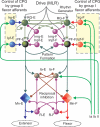
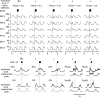
A computational model of the mammalian spinal cord circuitry incorporating a two-level central pattern generator (CPG) with separate half-centre rhythm generator (RG) and pattern formation (PF) networks has been developed from observations obtained during fictive locomotion in decerebrate cats. Sensory afferents have been incorporated in the model to study the effects of afferent stimulation on locomotor phase switching and step cycle period and on the firing patterns of flexor and extensor motoneurones. Here we show that this CPG structure can be integrated with reflex circuits to reproduce the reorganization of group I reflex pathways occurring during locomotion. During the extensor phase of fictive locomotion, activation of extensor muscle group I afferents increases extensor motoneurone activity and prolongs the extensor phase. This extensor phase prolongation may occur with or without a resetting of the locomotor cycle, which (according to the model) depends on the degree to which sensory input affects the RG and PF circuits, respectively. The same stimulation delivered during flexion produces a temporary resetting to extension without changing the timing of following locomotor cycles. The model reproduces this behaviour by suggesting that this sensory input influences the PF network without affecting the RG. The model also suggests that the different effects of flexor muscle nerve afferent stimulation observed experimentally (phase prolongation versus resetting) result from opposing influences of flexor group I and II afferents on the PF and RG circuits controlling the activity of flexor and extensor motoneurones. The results of modelling provide insights into proprioceptive control of locomotion.
Figures





References
-
- Booth V, Rinzel J, Kiehn O. Compartmental model of vertebrate motoneurons for Ca2+-dependent spiking and plateau potentials under pharmacological treatment. J Neurophysiol. 1997;78:3371–3385. - PubMed
-
- Buford JA, Smith JL. Adaptive control for backward quadrupedal walking. III. Stumbling corrective reactions and cutaneous reflex sensitivity. J Neurophysiol. 1993;70:1102–1114. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

