Modelling spinal circuitry involved in locomotor pattern generation: insights from deletions during fictive locomotion
- PMID: 17008376
- PMCID: PMC1890439
- DOI: 10.1113/jphysiol.2006.118703
Modelling spinal circuitry involved in locomotor pattern generation: insights from deletions during fictive locomotion
Abstract
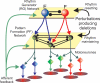
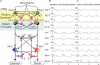
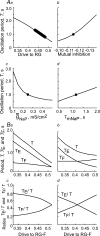
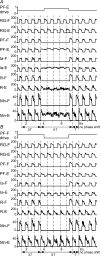
The mammalian spinal cord contains a locomotor central pattern generator (CPG) that can produce alternating rhythmic activity of flexor and extensor motoneurones in the absence of rhythmic input and proprioceptive feedback. During such fictive locomotor activity in decerebrate cats, spontaneous omissions of activity occur simultaneously in multiple agonist motoneurone pools for a number of cycles. During these 'deletions', antagonist motoneurone pools usually become tonically active but may also continue to be rhythmic. The rhythmic activity that re-emerges following a deletion is often not phase shifted. This suggests that some neuronal mechanism can maintain the locomotor period when motoneurone activity fails. To account for these observations, a simplified computational model of the spinal circuitry has been developed in which the locomotor CPG consists of two levels: a half-centre rhythm generator (RG) and a pattern formation (PF) network, with reciprocal inhibitory interactions between antagonist neural populations at each level. The model represents a network of interacting neural populations with single interneurones and motoneurones described in the Hodgkin-Huxley style. The model reproduces the range of locomotor periods and phase durations observed during real locomotion in adult cats and permits independent control of the level of motoneurone activity and of step cycle timing. By altering the excitability of neural populations within the PF network, the model can reproduce deletions in which motoneurone activity fails but the phase of locomotor oscillations is maintained. The model also suggests criteria for the functional identification of spinal interneurones involved in the mammalian locomotor pattern generation.
Figures





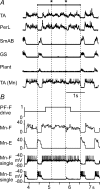
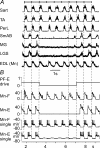
Comment in
-
Conceptualizing the mammalian locomotor central pattern generator with modelling.J Physiol. 2007 Apr 15;580(Pt. 2):363-4. doi: 10.1113/jphysiol.2007.129064. Epub 2007 Mar 1. J Physiol. 2007. PMID: 17331990 Free PMC article. Review. No abstract available.
Similar articles
-
Modelling spinal circuitry involved in locomotor pattern generation: insights from the effects of afferent stimulation.J Physiol. 2006 Dec 1;577(Pt 2):641-58. doi: 10.1113/jphysiol.2006.118711. Epub 2006 Sep 28. J Physiol. 2006. PMID: 17008375 Free PMC article.
-
Neuronal activity in the isolated mouse spinal cord during spontaneous deletions in fictive locomotion: insights into locomotor central pattern generator organization.J Physiol. 2012 Oct 1;590(19):4735-59. doi: 10.1113/jphysiol.2012.240895. Epub 2012 Aug 6. J Physiol. 2012. PMID: 22869012 Free PMC article.
-
Modeling the mammalian locomotor CPG: insights from mistakes and perturbations.Prog Brain Res. 2007;165:235-53. doi: 10.1016/S0079-6123(06)65015-2. Prog Brain Res. 2007. PMID: 17925250 Free PMC article. Review.
-
Deletions of rhythmic motoneuron activity during fictive locomotion and scratch provide clues to the organization of the mammalian central pattern generator.J Neurophysiol. 2005 Aug;94(2):1120-32. doi: 10.1152/jn.00216.2005. Epub 2005 May 4. J Neurophysiol. 2005. PMID: 15872066
-
Chapter 2--the spinal generation of phases and cycle duration.Prog Brain Res. 2011;188:15-29. doi: 10.1016/B978-0-444-53825-3.00007-3. Prog Brain Res. 2011. PMID: 21333800 Review.
Cited by
-
Motoneuronal and muscle synergies involved in cat hindlimb control during fictive and real locomotion: a comparison study.J Neurophysiol. 2012 Apr;107(8):2057-71. doi: 10.1152/jn.00865.2011. Epub 2011 Dec 21. J Neurophysiol. 2012. PMID: 22190626 Free PMC article.
-
Explicit Control of Step Timing During Split-Belt Walking Reveals Interdependent Recalibration of Movements in Space and Time.Front Hum Neurosci. 2019 Jul 3;13:207. doi: 10.3389/fnhum.2019.00207. eCollection 2019. Front Hum Neurosci. 2019. PMID: 31333429 Free PMC article.
-
Locomotor-related V3 interneurons initiate and coordinate muscles spasms after spinal cord injury.J Neurophysiol. 2019 Apr 1;121(4):1352-1367. doi: 10.1152/jn.00776.2018. Epub 2019 Jan 9. J Neurophysiol. 2019. PMID: 30625014 Free PMC article.
-
Persistent sodium currents participate in fictive locomotion generation in neonatal mouse spinal cord.J Neurosci. 2007 Apr 25;27(17):4507-18. doi: 10.1523/JNEUROSCI.0124-07.2007. J Neurosci. 2007. PMID: 17460064 Free PMC article.
-
Adaptive Control Strategies for Interlimb Coordination in Legged Robots: A Review.Front Neurorobot. 2017 Aug 23;11:39. doi: 10.3389/fnbot.2017.00039. eCollection 2017. Front Neurorobot. 2017. PMID: 28878645 Free PMC article. Review.
References
-
- Beato M, Nistri A. Interaction between disinhibited bursting and fictive locomotor patterns in the rat isolated spinal cord. J Neurophysiol. 1999;82:2029–2038. - PubMed
-
- Booth V, Rinzel J, Kiehn O. Compartmental model of vertebrate motoneurons for Ca2+-dependent spiking and plateau potentials under pharmacological treatment. J Neurophysiol. 1997;78:3371–3385. - PubMed
-
- Brownstone RM, Jordan LM, Kriellaars DJ, Noga BR, Shefchyk SJ. On the regulation of repetitive firing in lumbar motoneurones during fictive locomotion in the cat. Exp Brain Res. 1992;90:441–455. - PubMed
-
- Burke RE, Degtyarenko AM, Simon ES. Patterns of locomotor drive to motoneurons and last-order interneurons: clues to the structure of the CPG. J Neurophysiol. 2001;86:447–462. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

