Reduced nonprotein thiols inhibit activation and function of MMP-9: implications for chemoprevention
- PMID: 17015178
- PMCID: PMC2405910
- DOI: 10.1016/j.freeradbiomed.2006.07.014
Reduced nonprotein thiols inhibit activation and function of MMP-9: implications for chemoprevention
Abstract
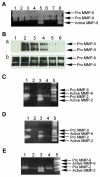
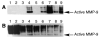
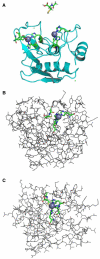
Clinical studies demonstrate a positive correlation between the extent of matrix metalloproteinase (MMP) activation and malignant progression of precancerous lesions. Therefore, identification of effective, well-tolerated MMP inhibitors represents a rational chemopreventive strategy. A variety of agents, including proteinases and thiol-oxidizing compounds, activate MMPs by initiating release of the propeptide's cysteine sulfur "blockage" of the MMP active site. Despite the importance of the propeptide's cysteine thiol in preserving MMP latency, limited studies have evaluated the effects of reduced thiols on MMP function. This study investigated the effects of two naturally occurring nonprotein thiols, i.e., glutathione (GSH) and N-acetylcysteine (NAC), on activation, function, and cellular-extracellular matrix interactions of the basement-membrane-degrading gelatinase, MMP-9. Our results reveal that NAC and GSH employ protein S-thiolation to inhibit organomercurial activation of pro-MMP-9. Gelatinase activity assays showed that GSH and NAC significantly inhibited MMP-9 but not MMP-2 function, implying isoform structural specificity. Immunoblot analyses, which suggested GSH interacts with MMP-9's active-site Zn, were corroborated by computational molecular modeling. Cell invasion assays revealed that NAC enhanced endostatin's ability to inhibit human cancer cell invasion. Collectively, these data demonstrate that nonprotein thiols suppress MMP-9 activation and function and introduce the prospect for their use in chemopreventive applications.
Figures


Similar articles
-
A new redox-dependent mechanism of MMP-1 activity control comprising reduced low-molecular-weight thiols and oxidizing radicals.J Mol Med (Berl). 2009 Mar;87(3):261-72. doi: 10.1007/s00109-008-0420-5. Epub 2008 Nov 26. J Mol Med (Berl). 2009. PMID: 19034402
-
Thiol supplementation inhibits metalloproteinase activity independent of glutathione status.Biochem Biophys Res Commun. 2007 Nov 23;363(3):651-5. doi: 10.1016/j.bbrc.2007.09.018. Epub 2007 Sep 18. Biochem Biophys Res Commun. 2007. PMID: 17900531
-
The inhibition effect of non-protein thiols on dentinal matrix metalloproteinase activity and HEMA cytotoxicity.J Dent. 2014 Mar;42(3):312-8. doi: 10.1016/j.jdent.2013.11.023. Epub 2013 Dec 4. J Dent. 2014. PMID: 24316344
-
Inhibitors of gelatinases (MMP-2 and MMP-9) for the management of hematological malignancies.Eur J Med Chem. 2021 Nov 5;223:113623. doi: 10.1016/j.ejmech.2021.113623. Epub 2021 Jun 12. Eur J Med Chem. 2021. PMID: 34157437 Review.
-
Physiological Properties, Functions, and Trends in the Matrix Metalloproteinase Inhibitors in Inflammation-Mediated Human Diseases.Curr Med Chem. 2023;30(18):2075-2112. doi: 10.2174/0929867329666220823112731. Curr Med Chem. 2023. PMID: 36017851 Review.
Cited by
-
Human head and neck squamous cell carcinoma cells are both targets and effectors for the angiogenic cytokine, VEGF.J Cell Biochem. 2008 Dec 1;105(5):1202-10. doi: 10.1002/jcb.21920. J Cell Biochem. 2008. PMID: 18802921 Free PMC article.
-
Phytochemical Profiling and Anti-Fibrotic Activities of the Gemmotherapy Bud Extract of Corylus avellana in a Model of Liver Fibrosis on Diabetic Mice.Biomedicines. 2023 Jun 20;11(6):1771. doi: 10.3390/biomedicines11061771. Biomedicines. 2023. PMID: 37371866 Free PMC article.
-
Glutathione has cell protective and anti-catabolic effects in articular cartilage without impairing the chondroanabolic phenotype.Heliyon. 2024 Nov 13;10(22):e40368. doi: 10.1016/j.heliyon.2024.e40368. eCollection 2024 Nov 30. Heliyon. 2024. PMID: 39624277 Free PMC article.
-
Diverse roles of matrix metalloproteinases and tissue inhibitors of metalloproteinases in neuroinflammation and cerebral ischemia.Neuroscience. 2009 Feb 6;158(3):983-94. doi: 10.1016/j.neuroscience.2008.06.025. Epub 2008 Jun 19. Neuroscience. 2009. PMID: 18621108 Free PMC article. Review.
-
Exogenous coenzyme Q10 modulates MMP-2 activity in MCF-7 cell line as a breast cancer cellular model.Nutr J. 2010 Nov 30;9:62. doi: 10.1186/1475-2891-9-62. Nutr J. 2010. PMID: 21118526 Free PMC article.
References
-
- Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function and biochemistry. Circ. Res. 2003;92:827–839. - PubMed
-
- Westermark J, Kahari VM. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J. 1999;13:781–792. - PubMed
-
- Blavier L, Henriet P, Imren S, Declerck YA. Tissue inhibitors of matrix metalloproteinases in cancer. Ann. N.Y. Acad. Sci. 1999;878:108–119. - PubMed
-
- Curran S, Marray GI. Matrix metalloproteinases in tumour invasion and metastasis. J. Pathol. 1999;189:300–308. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

