The ADP-stimulated NADPH oxidase activates the ASK-1/MKK4/JNK pathway in alveolar macrophages
- PMID: 17015265
- PMCID: PMC2713795
- DOI: 10.1080/10715760600758514
The ADP-stimulated NADPH oxidase activates the ASK-1/MKK4/JNK pathway in alveolar macrophages
Abstract
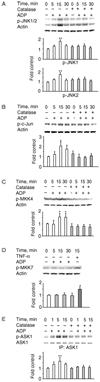
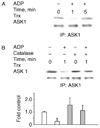
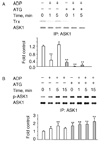
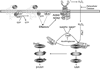
The role of H2O2 as a second messenger in signal transduction pathways is well established. We show here that the NADPH oxidase-dependent production of O2*(-) and H2O2 or respiratory burst in alveolar macrophages (AM) (NR8383 cells) is required for ADP-stimulated c-Jun phosphorylation and the activation of JNK1/2, MKK4 (but not MKK7) and apoptosis signal-regulating kinase-1 (ASK1). ASK1 binds only to the reduced form of thioredoxin (Trx). ADP induced the dissociation of ASK1/Trx complex and thus resulted in ASK1 activation, as assessed by phosphorylation at Thr845, which was enhanced after treatment with aurothioglucose (ATG), an inhibitor of Trx reductase. While dissociation of the complex implies Trx oxidation, protein electrophoretic mobility shift assay detected oxidation of Trx only after bolus H2O2 but not after ADP stimulation. These results demonstrate that the ADP-stimulated respiratory burst activated the ASK1-MKK4-JNK1/c-Jun signaling pathway in AM and suggest that transient and localized oxidation of Trx by the NADPH oxidase-mediated generation of H2O2 may play a critical role in ASK1 activation and the inflammatory response.
Figures

Similar articles
-
Apoptosis signal-regulating kinase 1-thioredoxin complex dissociation by capsaicin causes pancreatic tumor growth suppression by inducing apoptosis.Antioxid Redox Signal. 2012 Nov 15;17(10):1417-32. doi: 10.1089/ars.2011.4369. Epub 2012 Jun 11. Antioxid Redox Signal. 2012. PMID: 22530568 Free PMC article.
-
ADP stimulates the respiratory burst without activation of ERK and AKT in rat alveolar macrophages.Free Radic Biol Med. 2001 Sep 1;31(5):679-87. doi: 10.1016/s0891-5849(01)00630-x. Free Radic Biol Med. 2001. PMID: 11522453
-
Apoptosis signal-regulating kinase 1 activation by Nox1-derived oxidants is required for TNFα receptor endocytosis.Am J Physiol Heart Circ Physiol. 2019 Jun 1;316(6):H1528-H1537. doi: 10.1152/ajpheart.00741.2018. Epub 2019 Mar 29. Am J Physiol Heart Circ Physiol. 2019. PMID: 30925081 Free PMC article.
-
Macrophage signaling and respiratory burst.Immunol Res. 2002;26(1-3):95-105. doi: 10.1385/IR:26:1-3:095. Immunol Res. 2002. PMID: 12403349 Review.
-
Structural aspects of protein kinase ASK1 regulation.Adv Biol Regul. 2017 Dec;66:31-36. doi: 10.1016/j.jbior.2017.10.002. Epub 2017 Oct 16. Adv Biol Regul. 2017. PMID: 29066278 Review.
Cited by
-
Tumor necrosis factor-alpha and apoptosis signal-regulating kinase 1 control reactive oxygen species release, mitochondrial autophagy, and c-Jun N-terminal kinase/p38 phosphorylation during necrotizing enterocolitis.Oxid Med Cell Longev. 2009 Nov-Dec;2(5):297-306. doi: 10.4161/oxim.2.5.9541. Oxid Med Cell Longev. 2009. PMID: 20716917 Free PMC article.
-
Effect of hexane fraction of leaves of Cinnamomum tamala Linn on macrophage functions.Inflammopharmacology. 2010 Jun;18(3):147-54. doi: 10.1007/s10787-009-0029-5. Epub 2010 Jan 14. Inflammopharmacology. 2010. PMID: 20069379
-
An overview of mechanisms of redox signaling.J Mol Cell Cardiol. 2014 Aug;73:2-9. doi: 10.1016/j.yjmcc.2014.01.018. Epub 2014 Feb 8. J Mol Cell Cardiol. 2014. PMID: 24512843 Free PMC article. Review.
-
Redox-based regulation of signal transduction: principles, pitfalls, and promises.Free Radic Biol Med. 2008 Jul 1;45(1):1-17. doi: 10.1016/j.freeradbiomed.2008.03.011. Epub 2008 Mar 27. Free Radic Biol Med. 2008. PMID: 18423411 Free PMC article. Review.
-
Use and abuse of exogenous H2O2 in studies of signal transduction.Free Radic Biol Med. 2007 Apr 1;42(7):926-32. doi: 10.1016/j.freeradbiomed.2007.01.011. Epub 2007 Jan 12. Free Radic Biol Med. 2007. PMID: 17349920 Free PMC article. Review.
References
-
- Nauseef WM. Assembly of the phagocyte NADPH oxidase. Histochem Cell Biol. 2004 - PubMed
-
- Lambeth JD. NOX enzymes and the biology of reactive oxygen. Nat Rev Immunol. 2004;4:181–189. - PubMed
-
- Forman HJ, Torres M. Signaling by the respiratory burst in macrophages. IUBMB Life. 2001;51:365–371. - PubMed
-
- Simeonova PP, Luster MI. Iron and reactive oxygen species in the asbestos-induced tumor necrosis factor-alpha response from alveolar macrophages. Am J Respir Cell Mol Biol. 1995;12:676–683. - PubMed
-
- Gossart S, Cambon C, Orfila C, Seguelas M, Lepert J, Rami J, Carre P, Pipy B. Reactive oxygen intermediates as regulators of TNF-a production in rat lung inflammation induced by silica. J Immunol. 1996;156:1540–1548. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
