Functional role of GABAergic innervation of the cochlea: phenotypic analysis of mice lacking GABA(A) receptor subunits alpha 1, alpha 2, alpha 5, alpha 6, beta 2, beta 3, or delta
- PMID: 17021187
- PMCID: PMC1806703
- DOI: 10.1523/JNEUROSCI.2395-06.2006
Functional role of GABAergic innervation of the cochlea: phenotypic analysis of mice lacking GABA(A) receptor subunits alpha 1, alpha 2, alpha 5, alpha 6, beta 2, beta 3, or delta
Abstract
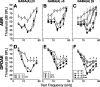
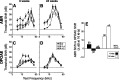
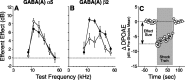
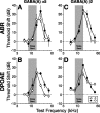
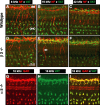
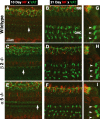
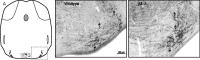
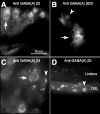
The olivocochlear efferent system is both cholinergic and GABAergic and innervates sensory cells and sensory neurons of the inner ear. Cholinergic effects on cochlear sensory cells are well characterized, both in vivo and in vitro; however, the robust GABAergic innervation is poorly understood. To explore the functional roles of GABA in the inner ear, we characterized the cochlear phenotype of seven mouse lines with targeted deletion of a GABA(A) receptor subunit (alpha1, alpha2, alpha5, alpha6, beta2, beta3, or delta). Four of the lines (alpha1, alpha2, alpha6, and delta) were normal: there was no cochlear histopathology, and cochlear responses suggested normal function of hair cells, afferent fibers, and efferent feedback. The other three lines (alpha5, beta2, and beta3) showed threshold elevations indicative of outer hair cell dysfunction. Alpha5 and beta2 lines also showed decreased effects of efferent bundle activation, associated with decreased density of efferent terminals on outer hair cells: although the onset of this degeneration was later in alpha5 (>6 weeks) than beta2 (<6 weeks), both lines shows normal efferent development (up to 3 weeks). Two lines (beta2 and beta3) showed signs of neuropathy, either decreased density of afferent innervation (beta3) or decreased neural responses without concomitant attenuation of hair cell responses (beta2). One of the lines (beta2) showed a clear sexual dimorphism in cochlear phenotype. Results suggest that the GABAergic component of the olivocochlear system contributes to the long-term maintenance of hair cells and neurons in the inner ear.
Figures



Similar articles
-
Loss of GABAB receptors in cochlear neurons: threshold elevation suggests modulation of outer hair cell function by type II afferent fibers.J Assoc Res Otolaryngol. 2009 Mar;10(1):50-63. doi: 10.1007/s10162-008-0138-7. Epub 2008 Oct 17. J Assoc Res Otolaryngol. 2009. PMID: 18925381 Free PMC article.
-
Immunohistochemical distribution of 10 GABAA receptor subunits in the forebrain of the rhesus monkey Macaca mulatta.J Comp Neurol. 2020 Oct 15;528(15):2551-2568. doi: 10.1002/cne.24910. Epub 2020 Apr 3. J Comp Neurol. 2020. PMID: 32220012 Free PMC article.
-
[Detection of GABA(A) receptor mRNA in cochlear tissue. An in situ hybridization study].HNO. 1995 Jan;43(1):12-8. HNO. 1995. PMID: 7890545 German.
-
gamma-Aminobutyric acid A receptor subunit mutant mice: new perspectives on alcohol actions.Biochem Pharmacol. 2004 Oct 15;68(8):1581-602. doi: 10.1016/j.bcp.2004.07.023. Biochem Pharmacol. 2004. PMID: 15451402 Review.
-
[Speech perception and otoacoustic emissions by pre-processing sound in the inner ear].HNO. 1992 Apr;40(4):111-22. HNO. 1992. PMID: 1318292 Review. German.
Cited by
-
The alpha1 subunit of the GABA(A) receptor modulates fear learning and plasticity in the lateral amygdala.Front Behav Neurosci. 2009 Oct 20;3:37. doi: 10.3389/neuro.08.037.2009. eCollection 2009. Front Behav Neurosci. 2009. PMID: 19876409 Free PMC article.
-
Monoallelic loss of the F-actin-binding protein radixin facilitates startle reactivity and pre-pulse inhibition in mice.Front Cell Dev Biol. 2022 Nov 28;10:987691. doi: 10.3389/fcell.2022.987691. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36518539 Free PMC article.
-
Hearing and vestibular deficits in the Coch(-/-) null mouse model: comparison to the Coch(G88E/G88E) mouse and to DFNA9 hearing and balance disorder.Hear Res. 2011 Feb;272(1-2):42-8. doi: 10.1016/j.heares.2010.11.002. Epub 2010 Nov 10. Hear Res. 2011. PMID: 21073934 Free PMC article.
-
Subtype selectivity of α+β- site ligands of GABAA receptors: identification of the first highly specific positive modulators at α6β2/3γ2 receptors.Br J Pharmacol. 2013 May;169(2):384-99. doi: 10.1111/bph.12153. Br J Pharmacol. 2013. PMID: 23472935 Free PMC article.
-
Activation of GABAB receptors results in excitatory modulation of calyx terminals in rat semicircular canal cristae.J Neurophysiol. 2020 Sep 1;124(3):962-972. doi: 10.1152/jn.00243.2020. Epub 2020 Aug 20. J Neurophysiol. 2020. PMID: 32816581 Free PMC article.
References
-
- Arnold T, Oestreicher E, Ehrenberger K, Felix D. GABA(A) receptor modulates the activity of inner hair cell afferents in guinea pig cochlea. Hear Res. 1998;125:147–153. - PubMed
-
- Babot Z, Cristofol R, Sunol C. Excitotoxic death induced by released glutamate in depolarized primary cultures of mouse cerebellar granule cells is dependent on GABAA receptors and niflumic acid-sensitive chloride channels. Eur J Neurosci. 2005;21:103–112. - PubMed
-
- Batta TJ, Panyi G, Szucs A, Sziklai I. Regulation of the lateral wall stiffness by acetylcholine and GABA in the outer hair cells of the guinea pig. Eur J Neurosci. 2004;20:3364–3370. - PubMed
-
- Bobbin RP, Bledsoe SC, Jr, Winbery S, Ceasar G, Jenison GL. Comparative actions of GABA and acetylcholine on the Xenopus laevis lateral line. Comp Biochem Physiol C. 1985;80:313–318. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
