Mitosis-specific mechanosensing and contractile-protein redistribution control cell shape
- PMID: 17027494
- PMCID: PMC2474462
- DOI: 10.1016/j.cub.2006.08.027
Mitosis-specific mechanosensing and contractile-protein redistribution control cell shape
Abstract
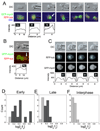
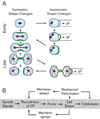
Because cell-division failure is deleterious, promoting tumorigenesis in mammals, cells utilize numerous mechanisms to control their cell-cycle progression. Though cell division is considered a well-ordered sequence of biochemical events, cytokinesis, an inherently mechanical process, must also be mechanically controlled to ensure that two equivalent daughter cells are produced with high fidelity. Given that cells respond to their mechanical environment, we hypothesized that cells utilize mechanosensing and mechanical feedback to sense and correct shape asymmetries during cytokinesis. Because the mitotic spindle and myosin II are vital to cell division, we explored their roles in responding to shape perturbations during cell division. We demonstrate that the contractile proteins myosin II and cortexillin I redistribute in response to intrinsic and externally induced shape asymmetries. In early cytokinesis, mechanical load overrides spindle cues and slows cytokinesis progression while contractile proteins accumulate and correct shape asymmetries. In late cytokinesis, mechanical perturbation also directs contractile proteins but without apparently disrupting cytokinesis. Significantly, this response only occurs during anaphase through cytokinesis, does not require microtubules, and is independent of spindle orientation, but is dependent on myosin II. Our data provide evidence for a mechanosensory system that directs contractile proteins to regulate cell shape during mitosis.
Figures


References
-
- Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D. Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature. 2005;437:1043–1047. - PubMed
-
- Norden C, Mendoza M, Dobbelaere J, Kotwaliwale CV, Biggins S, Barral Y. The nocut pathway links completion of cytokinesis to spindle midzone function to prevent chromosome breakage. Cell. 2006;125:85–98. - PubMed
-
- Wu J-Q, Kuhn JR, Kovar DR, Pollard TD. Spatial and temporal pathway for assembly and constriction of the contractile ring in fission yeast cytokinesis. Dev. Cell. 2003;5:723–734. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

