A seven-helix coiled coil
- PMID: 17030805
- PMCID: PMC1622844
- DOI: 10.1073/pnas.0604871103
A seven-helix coiled coil
Abstract
Coiled-coil proteins contain a characteristic seven-residue sequence repeat whose positions are designated a to g. The interacting surface between alpha-helices in a classical coiled coil is formed by interspersing nonpolar side chains at the a and d positions with hydrophilic residues at the flanking e and g positions. To explore how the chemical nature of these core amino acids dictates the overall coiled-coil architecture, we replaced all eight e and g residues in the GCN4 leucine zipper with nonpolar alanine side chains. Surprisingly, the alanine-containing mutant forms a stable alpha-helical heptamer in aqueous solution. The 1.25-A resolution crystal structure of the heptamer reveals a parallel seven-stranded coiled coil enclosing a large tubular channel with an unusual heptad register shift between adjacent staggered helices. The overall geometry comprises two interleaved hydrophobic helical screws of interacting cross-sectional a and d layers that have not been seen before. Moreover, asparagines at the a positions play an essential role in heptamer formation by participating in a set of buried interhelix hydrogen bonds. These results demonstrate that heptad repeats containing four hydrophobic positions can direct assembly of complex, higher-order coiled-coil structures with rich diversity for close packing of alpha-helices.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


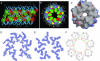

References
-
- Cohen C, Parry DA. Science. 1994;263:488–489. - PubMed
-
- Lupas AN, Gruber M. Adv Protein Chem. 2005;70:37–78. - PubMed
-
- Kohn WD, Mant CT, Hodges RS. J Biol Chem. 1997;272:2583–2586. - PubMed
-
- Betz SF, Bryson JW, DeGrado WF. Curr Opin Struct Biol. 1995;5:457–463. - PubMed
-
- McLachlan AD, Stewart M. J Mol Biol. 1975;98:293–304. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

