Predominant alpha2/beta2/gamma3 AMPK activation during exercise in human skeletal muscle
- PMID: 17038425
- PMCID: PMC1890393
- DOI: 10.1113/jphysiol.2006.120972
Predominant alpha2/beta2/gamma3 AMPK activation during exercise in human skeletal muscle
Abstract
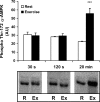
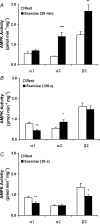
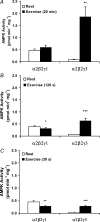
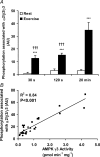
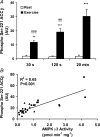
5'AMP-activated protein kinase (AMPK) is a key regulator of cellular metabolism and is regulated in muscle during exercise. We have previously established that only three of 12 possible AMPK alpha/beta/gamma-heterotrimers are present in human skeletal muscle. Previous studies describe discrepancies between total AMPK activity and regulation of its target acetyl-CoA-carboxylase (ACC)beta. Also, exercise training decreases expression of the regulatory gamma3 AMPK subunit and attenuates alpha2 AMPK activity during exercise. We hypothesize that these observations reflect a differential regulation of the AMPK heterotrimers. We provide evidence here that only the alpha2/beta2/gamma3 subunit is phosphorylated and activated during high-intensity exercise in vivo. The activity associated with the remaining two AMPK heterotrimers, alpha1/beta2/gamma1 and alpha2/beta2/gamma1, is either unchanged (20 min, 80% maximal oxygen uptake ) or decreased (30 or 120 s sprint-exercise). The differential activity of the heterotrimers leads to a total alpha-AMPK activity, that is decreased (30 s trial), unchanged (120 s trial) and increased (20 min trial). AMPK activity associated with the alpha2/beta2/gamma3 heterotrimer was strongly correlated to gamma3-associated alpha-Thr-172 AMPK phosphorylation (r(2) = 0.84, P < 0.001) and to ACCbeta Ser-221 phosphorylation (r(2) = 0.65, P < 0.001). These data single out the alpha2/beta2/gamma3 heterotrimer as an important actor in exercise-regulated AMPK signalling in human skeletal muscle, probably mediating phosphorylation of ACCbeta.
Figures
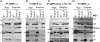
References
-
- Akerstrom TC, Birk JB, Klein DK, Erikstrup C, Plomgaard P, Pedersen BK, Wojtaszewski J. Oral glucose ingestion attenuates exercise-induced activation of 5′-AMP-activated protein kinase in human skeletal muscle. Biochem Biophys Res Commun. 2006;342:949–955. - PubMed
-
- Barnes BR, Marklund S, Steiler TL, Walter M, Hjalm G, Amarger V, Mahlapuu M, Leng Y, Johansson C, Galuska D, Lindgren K, Abrink M, Stapleton D, Zierath JR, Andersson L. The AMPK-gamma 3 isoform has a key role for carbohydrate and lipid metabolism in glycolytic skeletal muscle. J Biol Chem. 2004;279:38441–38447. - PubMed
-
- Bateman A. The structure of a domain common to archaebacteria and the homocystinuria disease protein. Trends Biochem Sci. 1997;22:12–13. - PubMed
-
- Chen ZP, McConell GK, Michell BJ, Snow RJ, Canny BJ, Kemp BE. AMPK signaling in contracting human skeletal muscle: acetyl-CoA carboxylase and NO synthase phosphorylation. Am J Physiol Endocrinol Metab. 2000;279:E1202–E1206. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

