Proteolytic processing is not essential for multiple functions of the Escherichia coli autotransporter adhesin involved in diffuse adherence (AIDA-I)
- PMID: 17041044
- PMCID: PMC1698232
- DOI: 10.1128/JB.00864-06
Proteolytic processing is not essential for multiple functions of the Escherichia coli autotransporter adhesin involved in diffuse adherence (AIDA-I)
Abstract
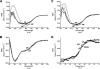
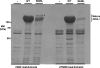
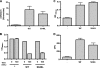
The Escherichia coli adhesin involved in diffuse adherence (AIDA-I), like many other autotransporter proteins, is released in the periplasm as a proprotein undergoing proteolytic processing after its translocation across the outer membrane. The proprotein is cleaved into a membrane-embedded fragment, AIDAc, and an extracellular fragment, the mature AIDA-I adhesin. The latter remains noncovalently associated with the outer membrane and can be released by heat treatment. The mechanism of cleavage of the proprotein and its role in the functionality of AIDA-I are not understood. Here, we show that cleavage is independent of the amount of AIDA-I in the outer membrane, suggesting an intramolecular autoproteolytic mechanism or a cleavage mediated by an unknown protease. We show that the two fragments, mature AIDA-I and AIDAc, can be cosolubilized and copurified in a folded and active conformation. We observed that the release by heat treatment results from the unfolding of AIDA-I and that the interaction of AIDA-I with AIDAc seems to be disturbed only by denaturation. We constructed an uncleavable point mutant of AIDA-I, where a serine of the cleavage site was changed into a leucine, and showed that adhesion, autoaggregation, and biofilm formation mediated by the mutant are indistinguishable from the wild-type levels. Lastly, we show that both proteins can mediate the invasion of cultured epithelial cells. Taken together, our experiments suggest that the proteolytic processing of AIDA-I plays a minor role in the functionality of this protein.
Figures




Similar articles
-
Autoprocessing of the Escherichia coli AIDA-I autotransporter: a new mechanism involving acidic residues in the junction region.J Biol Chem. 2009 Jun 19;284(25):17340-17351. doi: 10.1074/jbc.M109.010108. Epub 2009 Apr 27. J Biol Chem. 2009. PMID: 19398552 Free PMC article.
-
Barriers to folding of the transmembrane domain of the Escherichia coli autotransporter adhesin involved in diffuse adherence.Biochemistry. 2005 Mar 22;44(11):4533-45. doi: 10.1021/bi0475121. Biochemistry. 2005. PMID: 15766284
-
Contribution of AIDA-I to the pathogenicity of a porcine diarrheagenic Escherichia coli and to intestinal colonization through biofilm formation in pigs.Vet Microbiol. 2007 Mar 10;120(3-4):308-19. doi: 10.1016/j.vetmic.2006.10.035. Epub 2006 Nov 30. Vet Microbiol. 2007. PMID: 17140750
-
Self-associating autotransporters, SAATs: functional and structural similarities.Int J Med Microbiol. 2006 Aug;296(4-5):187-95. doi: 10.1016/j.ijmm.2005.10.002. Int J Med Microbiol. 2006. PMID: 16600681 Review.
-
Autotransporter Adhesins in Escherichia coli Pathogenesis.Proteomics. 2017 Dec;17(23-24). doi: 10.1002/pmic.201600431. Epub 2017 Oct 12. Proteomics. 2017. PMID: 28665015 Review.
Cited by
-
XatA, an AT-1 autotransporter important for the virulence of Xylella fastidiosa Temecula1.Microbiologyopen. 2012 Mar;1(1):33-45. doi: 10.1002/mbo3.6. Microbiologyopen. 2012. PMID: 22950010 Free PMC article.
-
Type V Secretion Systems: An Overview of Passenger Domain Functions.Front Microbiol. 2019 May 31;10:1163. doi: 10.3389/fmicb.2019.01163. eCollection 2019. Front Microbiol. 2019. PMID: 31214135 Free PMC article. Review.
-
Characterization of Shiga Toxin Subtypes and Virulence Genes in Porcine Shiga Toxin-Producing Escherichia coli.Front Microbiol. 2016 Apr 21;7:574. doi: 10.3389/fmicb.2016.00574. eCollection 2016. Front Microbiol. 2016. PMID: 27148249 Free PMC article.
-
Of linkers and autochaperones: an unambiguous nomenclature to identify common and uncommon themes for autotransporter secretion.Mol Microbiol. 2015 Jan;95(1):1-16. doi: 10.1111/mmi.12838. Epub 2014 Nov 24. Mol Microbiol. 2015. PMID: 25345653 Free PMC article. Review.
-
The Bartonella autotransporter BafA activates the host VEGF pathway to drive angiogenesis.Nat Commun. 2020 Jul 16;11(1):3571. doi: 10.1038/s41467-020-17391-2. Nat Commun. 2020. PMID: 32678094 Free PMC article.
References
-
- Benz, I., and M. A. Schmidt. 1992. AIDA-I, the adhesin involved in diffuse adherence of the diarrhoeagenic Escherichia coli strain 2787 (O126:H27), is synthesized via a precursor molecule. Mol. Microbiol. 6:1539-1546. - PubMed
-
- Benz, I., and M. A. Schmidt. 2001. Glycosylation with heptose residues mediated by the aah gene product is essential for adherence of the AIDA-I adhesin. Mol. Microbiol. 40:1403-1413. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

