Pheromone-induced degradation of Ste12 contributes to signal attenuation and the specificity of developmental fate
- PMID: 17041188
- PMCID: PMC1694826
- DOI: 10.1128/EC.00270-06
Pheromone-induced degradation of Ste12 contributes to signal attenuation and the specificity of developmental fate
Abstract
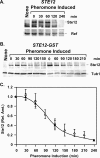
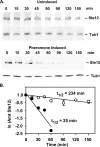
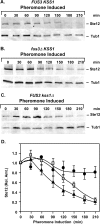
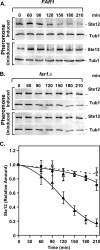
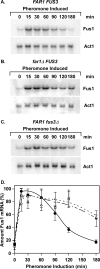
The Ste12 transcription factor of Saccharomyces cerevisiae regulates transcription programs controlling two different developmental fates. One is differentiation into a mating-competent form that occurs in response to mating pheromone. The other is the transition to a filamentous-growth form that occurs in response to nutrient deprivation. These two distinct roles for Ste12 make it a focus for studies into regulatory mechanisms that impart biological specificity. The transient signal characteristic of mating differentiation led us to test the hypothesis that regulation of Ste12 turnover might contribute to attenuation of the mating-specific transcription program and restrict activation of the filamentation program. We show that prolonged pheromone induction leads to ubiquitin-mediated destabilization and decreased amounts of Ste12. This depletion in pheromone-stimulated cultures is dependent on the mating-pathway-dedicated mitogen-activated protein kinase Fus3 and its target Cdc28 inhibitor, Far1. Attenuation of pheromone-induced mating-specific gene transcription (FUS1) temporally correlates with Ste12 depletion. This attenuation is abrogated in the deletion backgrounds (fus3Delta or far1Delta) where Ste12 is found to persist. Additionally, pheromone induces haploid invasion and filamentous-like growth instead of mating differentiation when Ste12 levels remain high. These observations indicate that loss of Ste12 reinforces the adaptive response to pheromone and contributes to the curtailing of a filamentation response.
Figures



Similar articles
-
Fus3-regulated Tec1 degradation through SCFCdc4 determines MAPK signaling specificity during mating in yeast.Cell. 2004 Dec 29;119(7):981-90. doi: 10.1016/j.cell.2004.11.053. Cell. 2004. PMID: 15620356
-
FUS3 phosphorylates multiple components of the mating signal transduction cascade: evidence for STE12 and FAR1.Mol Biol Cell. 1993 May;4(5):495-510. doi: 10.1091/mbc.4.5.495. Mol Biol Cell. 1993. PMID: 8334305 Free PMC article.
-
Regulation of the mating pheromone and invasive growth responses in yeast by two MAP kinase substrates.Curr Biol. 1997 Apr 1;7(4):228-38. doi: 10.1016/s0960-9822(06)00118-7. Curr Biol. 1997. PMID: 9094309
-
Ste12 and Ste12-like proteins, fungal transcription factors regulating development and pathogenicity.Eukaryot Cell. 2010 Apr;9(4):480-5. doi: 10.1128/EC.00333-09. Epub 2010 Feb 5. Eukaryot Cell. 2010. PMID: 20139240 Free PMC article. Review.
-
The pheromone signal pathway in Saccharomyces cerevisiae.Antonie Van Leeuwenhoek. 1992 Aug;62(1-2):95-108. doi: 10.1007/BF00584465. Antonie Van Leeuwenhoek. 1992. PMID: 1444339 Review.
Cited by
-
The same receptor, G protein, and mitogen-activated protein kinase pathway activate different downstream regulators in the alternative white and opaque pheromone responses of Candida albicans.Mol Biol Cell. 2008 Mar;19(3):957-70. doi: 10.1091/mbc.e07-07-0688. Epub 2007 Dec 27. Mol Biol Cell. 2008. PMID: 18162580 Free PMC article.
-
Nucleus-specific and cell cycle-regulated degradation of mitogen-activated protein kinase scaffold protein Ste5 contributes to the control of signaling competence.Mol Cell Biol. 2009 Jan;29(2):582-601. doi: 10.1128/MCB.01019-08. Epub 2008 Nov 10. Mol Cell Biol. 2009. PMID: 19001089 Free PMC article.
-
The regulation of filamentous growth in yeast.Genetics. 2012 Jan;190(1):23-49. doi: 10.1534/genetics.111.127456. Genetics. 2012. PMID: 22219507 Free PMC article. Review.
-
Pheromone- and RSP5-dependent ubiquitination of the G protein beta subunit Ste4 in yeast.J Biol Chem. 2011 Aug 5;286(31):27147-55. doi: 10.1074/jbc.M111.254193. Epub 2011 Jun 17. J Biol Chem. 2011. PMID: 21685393 Free PMC article.
-
Sumoylation of transcription factor Tec1 regulates signaling of mitogen-activated protein kinase pathways in yeast.PLoS One. 2009 Oct 14;4(10):e7456. doi: 10.1371/journal.pone.0007456. PLoS One. 2009. PMID: 19826484 Free PMC article.
References
-
- Bao, M. Z., M. A. Schwartz, G. T. Cantin, J. R. Yates III, and H. D. Madhani. 2004. Pheromone-dependent destruction of the Tec1 transcription factor is required for MAP kinase signaling specificity in yeast. Cell 119:991-1000. - PubMed
-
- Breitkreutz, A. 2002. Signal specificity in the budding yeast mating/filamentous MAPK pathway. University of Toronto, Toronto, Canada.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

