Serratia marcescens serralysin induces inflammatory responses through protease-activated receptor 2
- PMID: 17043106
- PMCID: PMC1828393
- DOI: 10.1128/IAI.01239-06
Serratia marcescens serralysin induces inflammatory responses through protease-activated receptor 2
Abstract
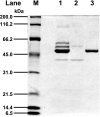
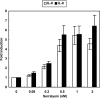
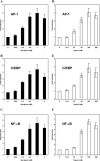
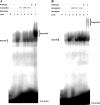
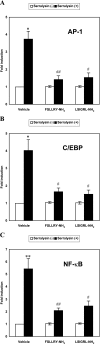
The Serratia marcescens-derived protease serralysin is considered to play an important role in the pathogenesis of infection. Protease-activated receptor 2 (PAR-2) is activated by trypsin and also several other trypsin-like serine proteases, leading to the modulation of inflammatory and immune responses. However, little is known about the activation of PAR-2 by bacterial proteases and its roles in bacterial infection. In this study, we investigated whether S. marcescens serralysin activates host inflammatory responses through PAR-2. Our results demonstrated that serralysin induces interleukin-6 (IL-6) and IL-8 mRNA expression in a human lung squamous cell carcinoma, EBC-l cells. In addition, serralysin activated activator protein 1 (AP-1)-, CCAAT/enhancer-binding protein (C/EBP)-, and nuclear factor-kappaB (NF-kappaB)-driven promoters in EBC-1 cells. An electrophoretic mobility shift assay showed that serralysin activates the binding of AP-1, C/EBPbeta, and NF-kappaB in the cells. Inactivation of serralysin resulted in the failure of transactivation of AP-1-, C/EBP-, and NF-kappaB-driven promoters in the cells. Furthermore, serralysin activated AP-1-, C/EBP-, and NF-kappaB-driven promoters via PAR-2 in HeLa cells. PAR-2 antagonist peptides decreased serralysin-induced transactivation of AP-1-, C/EBP-, and NF-kappaB-driven promoters in EBC-1 cells. Considered together, these results suggest that serralysin requires PAR-2 to activate the critical transcription factors AP-1, C/EBPbeta, and NF-kappaB for host inflammatory responses.
Figures


Similar articles
-
Transcriptional regulation of IL-8 by Staphylococcus aureus in human conjunctival cells involves activation of AP-1.Invest Ophthalmol Vis Sci. 2007 Jan;48(1):270-6. doi: 10.1167/iovs.06-0081. Invest Ophthalmol Vis Sci. 2007. PMID: 17197543
-
PAR-2 activation regulates IL-8 and GRO-alpha synthesis by NF-kappaB, but not RANTES, IL-6, eotaxin or TARC expression in nasal epithelium.Clin Exp Allergy. 2007 Jul;37(7):1009-22. doi: 10.1111/j.1365-2222.2007.02686.x. Clin Exp Allergy. 2007. PMID: 17581194
-
A novel secreted protease from Pseudomonas aeruginosa activates NF-kappaB through protease-activated receptors.Cell Microbiol. 2008 Jul;10(7):1491-504. doi: 10.1111/j.1462-5822.2008.01142.x. Epub 2008 Mar 10. Cell Microbiol. 2008. PMID: 18331590
-
Regulation of the human oxytocin receptor by nuclear factor-kappaB and CCAAT/enhancer-binding protein-beta.J Clin Endocrinol Metab. 2006 Jun;91(6):2317-26. doi: 10.1210/jc.2005-2649. Epub 2006 Mar 28. J Clin Endocrinol Metab. 2006. PMID: 16569740
-
Basic and translational research on proteinase-activated receptors: implication of proteinase/proteinase-activated receptor in gastrointestinal inflammation.J Pharmacol Sci. 2008 Dec;108(4):415-21. doi: 10.1254/jphs.08r31fm. J Pharmacol Sci. 2008. PMID: 19098387 Review.
Cited by
-
Gut Microbiota in Patients with Different Metabolic Statuses: Moscow Study.Microorganisms. 2018 Sep 25;6(4):98. doi: 10.3390/microorganisms6040098. Microorganisms. 2018. PMID: 30257444 Free PMC article.
-
Plasma amino acid profiles in dogs with inflammatory bowel disease.J Vet Intern Med. 2019 Jul;33(4):1602-1607. doi: 10.1111/jvim.15525. Epub 2019 May 20. J Vet Intern Med. 2019. PMID: 31111561 Free PMC article.
-
Xylitol Inhibits Growth and Blocks Virulence in Serratia marcescens.Microorganisms. 2021 May 18;9(5):1083. doi: 10.3390/microorganisms9051083. Microorganisms. 2021. PMID: 34070043 Free PMC article.
-
Protease-activated receptors (PARs): mechanisms of action and potential therapeutic modulators in PAR-driven inflammatory diseases.Thromb J. 2019 Mar 29;17:4. doi: 10.1186/s12959-019-0194-8. eCollection 2019. Thromb J. 2019. PMID: 30976204 Free PMC article. Review.
-
Protease-activated receptor 2 (PAR2) is upregulated by Acanthamoeba plasminogen activator (aPA) and induces proinflammatory cytokine in human corneal epithelial cells.Invest Ophthalmol Vis Sci. 2014 May 29;55(6):3912-21. doi: 10.1167/iovs.14-14486. Invest Ophthalmol Vis Sci. 2014. PMID: 24876278 Free PMC article.
References
-
- Adam, E., K. K. Hansen, O. F. Astudillo, L. Coulon, F. Bex, X. Duhant, E. Jaumotte, M. D. Hollenberg, and A. Jacquet. 2006. The house dust mite allergen Der p 1, unlike Der p 3, stimulates the expression of interleukin-8 in human airway epithelial cells via a proteinase-activated receptor-2-independent mechanism. J. Biol. Chem. 281:6910-6923. - PubMed
-
- Al-Ani, B., M. Saifeddine, S. J. Wijesuriya, and M. D. Hollenberg. 2002. Modified proteinase-activated receptor-1 and -2 derived peptides inhibit proteinase-activated receptor-2 activation by trypsin. J. Pharmacol. Exp. Ther. 300:702-708. - PubMed
-
- Albers, M. J., J. W. Mouton, and D. Tibboel. 2001. Colonization and infection by Serratia species in a paediatric surgical intensive care unit. J. Hosp. Infect. 48:7-12. - PubMed
-
- Asokananthan, N., P. T. Graham, D. J. Stewart, A. J. Bakker, K. A. Eidne, P. J. Thompson, and G. A. Stewart. 2002. House dust mite allergens induce proinflammatory cytokines from respiratory epithelial cells: the cysteine protease allergen, Der p 1, activates protease-activated receptor (PAR)-2 and inactivates PAR-1. J. Immunol. 169:4572-4578. - PubMed
-
- Aucken, H. M., and T. L. Pitt. 1998. Antibiotic resistance and putative virulence factors of Serratia marcescens with respect to O and K serotypes. J. Med. Microbiol. 47:1105-1113. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

