Effect of anaerobic growth on quinolone lethality with Escherichia coli
- PMID: 17043118
- PMCID: PMC1797672
- DOI: 10.1128/AAC.00739-06
Effect of anaerobic growth on quinolone lethality with Escherichia coli
Abstract
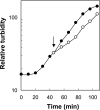
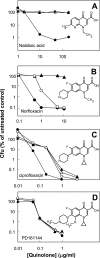
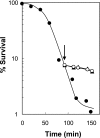
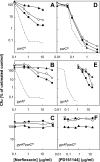
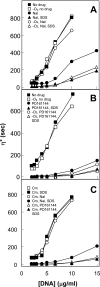
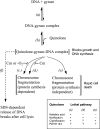
Quinolone activity against Escherichia coli was examined during aerobic growth, aerobic treatment with chloramphenicol, and anaerobic growth. Nalidixic acid, norfloxacin, ciprofloxacin, and PD161144 were lethal for cultures growing aerobically, and the bacteriostatic activity of each quinolone was unaffected by anaerobic growth. However, lethal activity was distinct for each quinolone with cells treated aerobically with chloramphenicol or grown anaerobically. Nalidixic acid failed to kill cells under both conditions; norfloxacin killed cells when they were grown anaerobically but not when they were treated with chloramphenicol; ciprofloxacin killed cells under both conditions but required higher concentrations than those required with cells grown aerobically; and PD161144, a C-8-methoxy fluoroquinolone, was equally lethal under all conditions. Following pretreatment with nalidixic acid, a shift to anaerobic conditions or the addition of chloramphenicol rapidly blocked further cell death. Formation of quinolone-gyrase-DNA complexes, observed as a sodium dodecyl sulfate (SDS)-dependent drop in cell lysate viscosity, occurred during aerobic and anaerobic growth and in the presence and in the absence of chloramphenicol. However, lethal chromosome fragmentation, detected as a drop in viscosity in the absence of SDS, occurred with nalidixic acid treatment only under aerobic conditions in the absence of chloramphenicol. With PD161144, chromosome fragmentation was detected when the cells were grown aerobically and anaerobically and in the presence and in the absence of chloramphenicol. Thus, all quinolones tested appear to form reversible bacteriostatic complexes containing broken DNA during aerobic growth, during anaerobic growth, and when protein synthesis is blocked; however, the ability to fragment chromosomes and to rapidly kill cells under these conditions depends on quinolone structure.
Figures
Similar articles
-
Lethal fragmentation of bacterial chromosomes mediated by DNA gyrase and quinolones.Mol Microbiol. 2006 Aug;61(3):810-25. doi: 10.1111/j.1365-2958.2006.05275.x. Epub 2006 Jun 27. Mol Microbiol. 2006. PMID: 16803589
-
DNA gyrase and topoisomerase IV on the bacterial chromosome: quinolone-induced DNA cleavage.J Mol Biol. 1996 May 17;258(4):627-37. doi: 10.1006/jmbi.1996.0274. J Mol Biol. 1996. PMID: 8636997
-
Lethal action of quinolones against a temperature-sensitive dnaB replication mutant of Escherichia coli.Antimicrob Agents Chemother. 2006 Jan;50(1):362-4. doi: 10.1128/AAC.50.1.362-364.2006. Antimicrob Agents Chemother. 2006. PMID: 16377712 Free PMC article.
-
Mechanisms of action of and resistance to ciprofloxacin.Am J Med. 1987 Apr 27;82(4A):12-20. Am J Med. 1987. PMID: 3034057 Review.
-
[The evolution of quinolone: from nalidixic acid to the quinolones of the third generation].Pharm Unserer Zeit. 2001;30(5):376-81. doi: 10.1002/1615-1003(200109)30:5<376::AID-PAUZ376>3.0.CO;2-O. Pharm Unserer Zeit. 2001. PMID: 11575173 Review. German. No abstract available.
Cited by
-
Antibiotic-persistent bacterial cells exhibiting low-level ROS are eradicated by ROS-independent membrane disruption.mBio. 2025 Aug 13;16(8):e0119925. doi: 10.1128/mbio.01199-25. Epub 2025 Jun 30. mBio. 2025. PMID: 40586599 Free PMC article.
-
Enhancement of the Bactericidal Effect of Antibiotics by Inhibition of Enzymes Involved in Production of Hydrogen Sulfide in Bacteria.Mol Biol. 2022;56(5):638-648. doi: 10.1134/S0026893322050120. Epub 2022 Oct 5. Mol Biol. 2022. PMID: 36217334 Free PMC article.
-
Quinolone-mediated bacterial death.Antimicrob Agents Chemother. 2008 Feb;52(2):385-92. doi: 10.1128/AAC.01617-06. Epub 2007 Aug 27. Antimicrob Agents Chemother. 2008. PMID: 17724149 Free PMC article. Review. No abstract available.
-
Antibiotic-Induced Bacterial Cell Death: A "Radical" Way of Dying?Curr Top Microbiol Immunol. 2025 Aug 9:10.1007/82_2024_284. doi: 10.1007/82_2024_284. Online ahead of print. Curr Top Microbiol Immunol. 2025. PMID: 40779246
-
Moving forward with reactive oxygen species involvement in antimicrobial lethality.J Antimicrob Chemother. 2015 Mar;70(3):639-42. doi: 10.1093/jac/dku463. Epub 2014 Nov 23. J Antimicrob Chemother. 2015. PMID: 25422287 Free PMC article. Review.
References
-
- Chen, C. R., M. Malik, M. Snyder, and K. Drlica. 1996. DNA gyrase and topoisomerase IV on the bacterial chromosome: quinolone-induced DNA cleavage. J. Mol. Biol. 258:627-637. - PubMed
-
- Chow, R., T. Dougherty, H. Fraimow, E. Bellin, and M. Miller. 1988. Association between early inhibition of DNA synthesis and the MICs and MBCs of carboxyquinolone antimicrobial agents for wild-type and mutant [gyrA nfxB(ompF) acrA] Escherichia coli K-12. Antimicrob. Agents Chemother. 32:1113-1118. - PMC - PubMed
-
- Cooper, M. A., J. M. Andrews, and R. Wise. 1991. Bactericidal activity of sparfloxacin and ciprofloxacin under anaerobic conditions. J. Antimicrob. Chemother. 28:399-405. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

