Recruitment of CD63 to Cryptococcus neoformans phagosomes requires acidification
- PMID: 17043215
- PMCID: PMC1635107
- DOI: 10.1073/pnas.0607528103
Recruitment of CD63 to Cryptococcus neoformans phagosomes requires acidification
Abstract
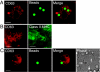
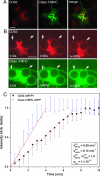
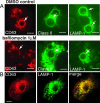
The subcellular localization of the cluster of differentiation 63 (CD63) tetraspanin and its interaction with the class II MHC antigen presentation pathway were examined in the context of phagocytosis by live cell imaging, by using monomeric red fluorescent protein-tagged mouse CD63 expressed in primary bone marrow-derived cell cultures. Upon phagocytosis of Cryptococcus neoformans and polystyrene beads, CD63 was recruited selectively to C. neoformans-containing phagosomes in a MyD88-independent acidification-dependent manner. Bead-containing phagosomes, within a C. neoformans-containing cell, acidified to a lesser extent and failed to recruit CD63 to a level detectable by microscopy. CD63 recruitment to yeast phagosomes occurred independently of class II MHC and LAMP-1. These observations indicate that the composition of distinct phagosomal compartments within the same cell is determined by phagosomal cargo and may affect the outcome of antigen processing and presentation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

