Assembly and structural characterization of an authentic complex between human follicle stimulating hormone and a hormone-binding ectodomain of its receptor
- PMID: 17045735
- PMCID: PMC2012943
- DOI: 10.1016/j.mce.2005.12.055
Assembly and structural characterization of an authentic complex between human follicle stimulating hormone and a hormone-binding ectodomain of its receptor
Abstract
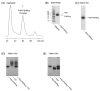
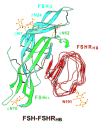
Follicle stimulating hormone (FSH) is secreted from the pituitary gland to regulate reproduction in vertebrates. FSH signals through a G-protein coupled receptor (FSHR) on the target cell surface. We describe here the strategy to produce a soluble FSH-FSHR complex that involves the co-secretion of a truncated FSHR ectodomain (FSHR(HB)) and a covalently linked FSHalphabeta heterodimer from baculovirus-infected insect cells. FSH binds to FSHR(HB) with a high affinity comparable to that for the full-length receptor. The crystal structure of the FSH-FSHR(HB) complex provides explanations for the high affinity and specificity of FSH interaction with FSHR, and it shows an unexpected dimerization of these complexes. Here we also compare the crystal structure with theoretical models of the FSH-FSHR-binding mode. We conclude that the FSH-FSHR(HB) structure gives an authentic representation of FSH binding to intact FSHR.
Figures


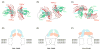

Similar articles
-
Structure of human follicle-stimulating hormone in complex with its receptor.Nature. 2005 Jan 20;433(7023):269-77. doi: 10.1038/nature03206. Nature. 2005. PMID: 15662415 Free PMC article.
-
Structure of follicle-stimulating hormone in complex with the entire ectodomain of its receptor.Proc Natl Acad Sci U S A. 2012 Jul 31;109(31):12491-6. doi: 10.1073/pnas.1206643109. Epub 2012 Jul 16. Proc Natl Acad Sci U S A. 2012. PMID: 22802634 Free PMC article.
-
Structural biology of glycoprotein hormones and their receptors: insights to signaling.Mol Cell Endocrinol. 2014 Jan 25;382(1):424-451. doi: 10.1016/j.mce.2013.08.021. Epub 2013 Aug 31. Mol Cell Endocrinol. 2014. PMID: 24001578 Review.
-
Identification of the sites of N-linked glycosylation on the follicle-stimulating hormone (FSH) receptor and assessment of their role in FSH receptor function.Mol Endocrinol. 1995 Feb;9(2):159-70. doi: 10.1210/mend.9.2.7776966. Mol Endocrinol. 1995. PMID: 7776966
-
Follicle-Stimulating Hormone Receptor: Advances and Remaining Challenges.Int Rev Cell Mol Biol. 2018;338:1-58. doi: 10.1016/bs.ircmb.2018.02.001. Epub 2018 Apr 5. Int Rev Cell Mol Biol. 2018. PMID: 29699689 Review.
Cited by
-
The heterodimeric glycoprotein hormone, GPA2/GPB5, regulates ion transport across the hindgut of the adult mosquito, Aedes aegypti.PLoS One. 2014 Jan 20;9(1):e86386. doi: 10.1371/journal.pone.0086386. eCollection 2014. PLoS One. 2014. PMID: 24466069 Free PMC article.
-
Misfolding Ectodomain Mutations of the Lutropin Receptor Increase Efficacy of Hormone Stimulation.Mol Endocrinol. 2016 Jan;30(1):62-76. doi: 10.1210/me.2015-1205. Epub 2015 Nov 10. Mol Endocrinol. 2016. PMID: 26554443 Free PMC article.
-
Discovery and Preclinical Development of Orally Active Small Molecules that Exhibit Highly Selective Follicle Stimulating Hormone Receptor Agonism.Front Pharmacol. 2021 Jan 14;11:602593. doi: 10.3389/fphar.2020.602593. eCollection 2020. Front Pharmacol. 2021. PMID: 33519465 Free PMC article.
-
The luteinizing hormone receptor: insights into structure-function relationships and hormone-receptor-mediated changes in gene expression in ovarian cancer cells.Mol Cell Endocrinol. 2010 Nov 25;329(1-2):47-55. doi: 10.1016/j.mce.2010.04.025. Epub 2010 May 2. Mol Cell Endocrinol. 2010. PMID: 20444430 Free PMC article. Review.
-
New Frontier in Glycoprotein Hormones and Their Receptors Structure-Function.Front Endocrinol (Lausanne). 2015 Oct 19;6:155. doi: 10.3389/fendo.2015.00155. eCollection 2015. Front Endocrinol (Lausanne). 2015. PMID: 26539160 Free PMC article. Review.
References
-
- Arnold CJ, Liu C, Lindau-Shepard B, Losavio ML, Patrascu MT, Dias JA. The human follitropin α-subunit C terminus collaborates with a β-subunit cystine noose and an α-subunit loop to assemble a receptor-binding domain competent for signal transduction. Biochemistry. 1998;37:1762–1768. - PubMed
-
- Ascoli M, Fanelli F, Segaloff DL. The lutropin/choriogonadotropin receptor, a 2002 perspective. Endocr Rev. 2002;23:141–174. - PubMed
-
- Bhowmick N, Huang J, Puett D, Isaacs NW, Lapthorn AJ. Determination of residues important in hormone binding to the extracellular domain of the luteinizing hormone/chorionic gonadotropin receptor by site-directed mutagenesis and modeling. Mol Endocrinol. 1996;10:1147–1159. - PubMed
-
- Bhowmick N, Narayan P, Puett D. Identification of ionizable amino acid residues on the extracellular domain of the lutropin receptor involved in ligand binding. Endocrinology. 1999;140:4558–4563. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

