The oncolytic effect in vivo of reovirus on tumour cells that have survived reovirus cell killing in vitro
- PMID: 17047650
- PMCID: PMC2360720
- DOI: 10.1038/sj.bjc.6603363
The oncolytic effect in vivo of reovirus on tumour cells that have survived reovirus cell killing in vitro
Abstract
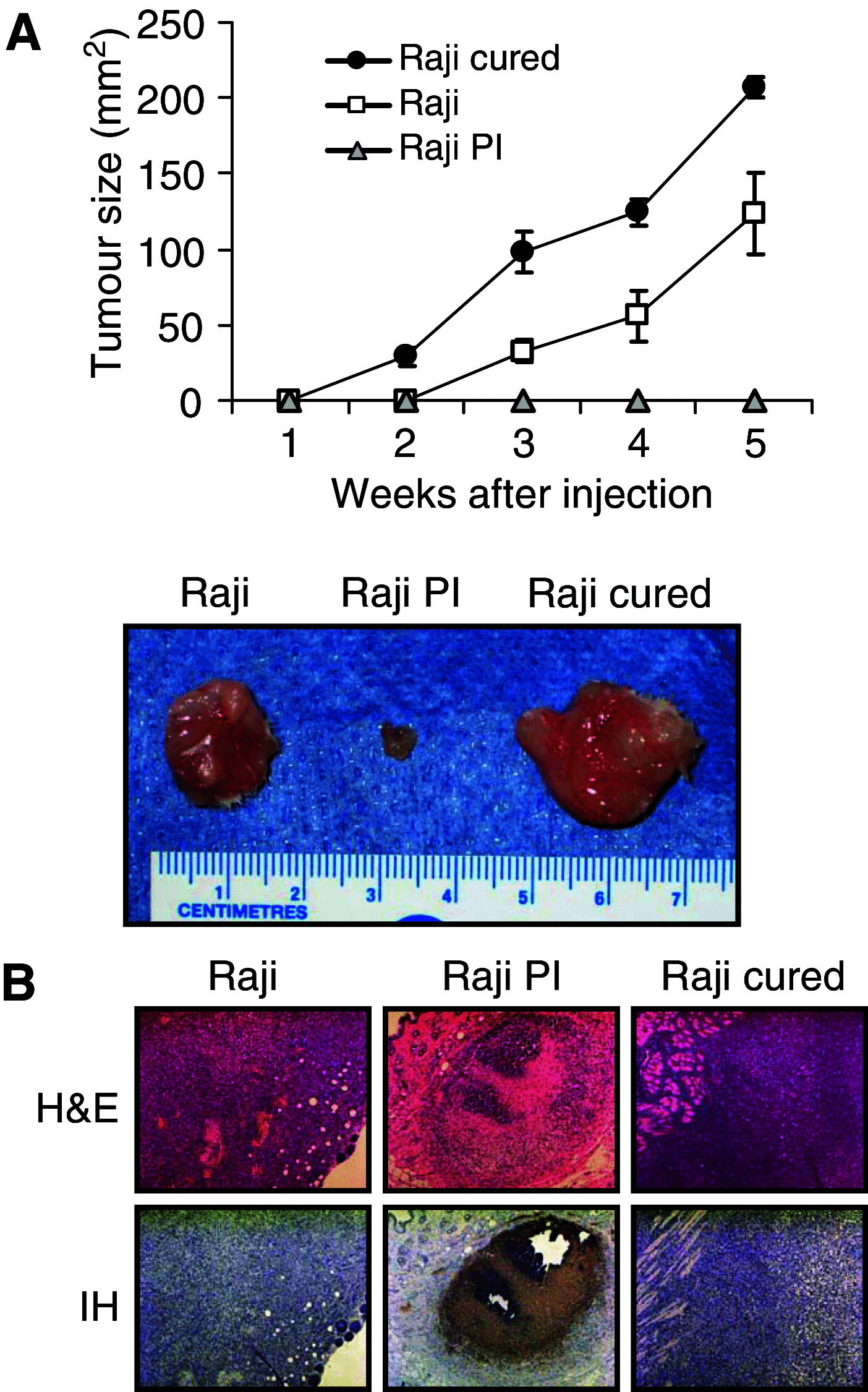
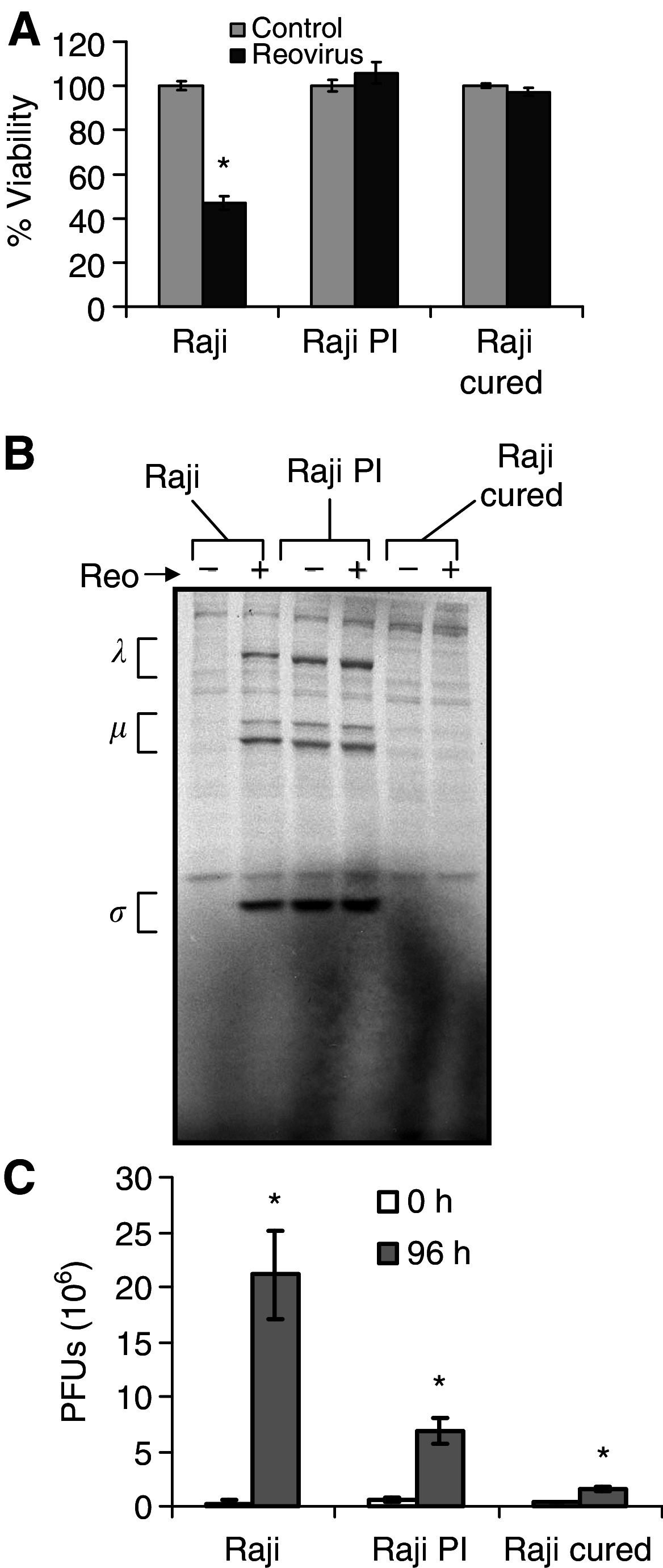
The use of oncolytic viruses has received considerable attention in recent years and many viruses have proved to be effective against a variety of cancer models and a few are currently being used in clinical trials. However, the possible emergence and outcome of virus-resistant tumour cells has not been addressed. We previously reported the effective use of reovirus against lymphoid malignancies, including the Burkitt's lymphoma cell line Raji. Here we isolated in vitro persistently infected (PI) Raji cells, and cells 'cured' of persistent reovirus infection ('cured' cells). Both PI and cured Raji cells resisted reovirus infection and cell killing in vitro. In vivo, the PI cells were non-tumorigenic in SCID mice, but cured cells regained the parental cells' ability to form tumours. Tumour xenografts from the cured cells, however, were highly susceptible to reovirus oncolysis in vivo. This susceptibility was due to the proteolytic environment within tumours that facilitates reovirus infection and cell killing. Our results show that persistent infection by reovirus impedes tumour development and that although PI cells cleared of reovirus are tumorigenic, they are killed upon rechallenge with reovirus. Both the PI and cured states are therefore not likely to be significant barriers to reovirus oncolytic therapy.
Figures
References
-
- Ahmed R, Fields BN (1982) Role of the S4 gene in the establishment of persistent reovirus infection in L cells. Cell 28: 605–612 - PubMed
-
- Ahmed R, Canning WM, Kauffman RS, Sharpe AH, Hallum JV, Fields BN (1981) Role of the host cell in persistent viral infection: coevolution of L cells and reovirus during persistent infection. Cell 25: 325–332 - PubMed
-
- Alain T, Hirasawa K, Pon KJ, Nishikawa SG, Urbanski SJ, Auer Y, Luider J, Martin A, Johnston RN, Janowska-Wieczorek A, Lee PW, Kossakowska AE (2002) Reovirus therapy of lymphoid malignancies. Blood 100: 4146–4153 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

