Cell-specific differential modulation of human trabecular meshwork cells by selective adenosine receptor agonists
- PMID: 17070802
- PMCID: PMC1764820
- DOI: 10.1016/j.exer.2006.09.007
Cell-specific differential modulation of human trabecular meshwork cells by selective adenosine receptor agonists
Abstract
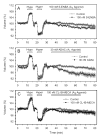
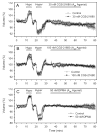
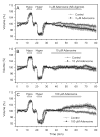
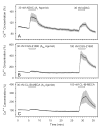
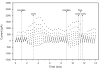
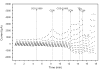
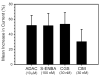
Activation of A1 and A2A subtype adenosine receptors (AR) likely exert opposing effects on outflow of aqueous humor, and thereby, on intraocular pressure. Selective agonists of adenosine receptor (AR) subtypes have previously been applied to trabecular meshwork (TM) and Schlemm's canal (SC) cells to identify the site(s) of differential purinergic modulation. However, the apparent changes in volume monitored by previously measuring projected cell area might have partially reflected cell contraction and relaxation. In addition, whole-cell current responses of the TM cells previously described were highly variable following application of selective A1, A2A and A3 agonists. The complexity of the electrophysiologic responses may have reflected cell heterogeneity of the populations harvested from collagenase digestion of TM explants. We now report measurements of TM-cell volume using calcein fluorescence quenching, an approach independent of contractile state. Furthermore, we have applied selective AR agonists to a uniform population of human TM cells, the hTM5 cell line. A1, but not A2A or A3, AR agonists triggered TM-cell shrinkage. Both A1 and A2A AR agonists produced reproducible increases in TM-cell whole-cell currents of similar magnitude. The results suggest that previous measurements of explant-derived TM cells may have reflected a range of responses from phenotypically different cell populations, and that the opposing effects of A1 and A2A agonists on outflow resistance are not likely to be mediated by actions on a single population of TM cells. These opposing effects might reflect AR responses by two or more subpopulations of TM cells, by TM and SC cells or by inner-wall SC cells, alone.
Figures

Similar articles
-
Common actions of adenosine receptor agonists in modulating human trabecular meshwork cell transport.J Membr Biol. 2003 May 15;193(2):121-36. doi: 10.1007/s00232-002-2013-5. J Membr Biol. 2003. PMID: 12879160
-
Differential P1-purinergic modulation of human Schlemm's canal inner-wall cells.Am J Physiol Cell Physiol. 2005 Apr;288(4):C784-94. doi: 10.1152/ajpcell.00333.2004. Epub 2004 Dec 8. Am J Physiol Cell Physiol. 2005. PMID: 15590899
-
Electron probe X-ray microanalysis of intact pathway for human aqueous humor outflow.Am J Physiol Cell Physiol. 2008 Nov;295(5):C1083-91. doi: 10.1152/ajpcell.340.2008. Epub 2008 Aug 27. Am J Physiol Cell Physiol. 2008. PMID: 18753314 Free PMC article.
-
Newer targets for modulation of intraocular pressure: focus on adenosine receptor signaling pathways.Expert Opin Ther Targets. 2014 May;18(5):527-39. doi: 10.1517/14728222.2014.888416. Epub 2014 Mar 1. Expert Opin Ther Targets. 2014. PMID: 24579961 Review.
-
Adenosine, adenosine receptors and glaucoma: an updated overview.Biochim Biophys Acta. 2013 Apr;1830(4):2882-90. doi: 10.1016/j.bbagen.2013.01.005. Epub 2013 Jan 15. Biochim Biophys Acta. 2013. PMID: 23328492 Review.
Cited by
-
Recent Patents on Emerging Therapeutics for the Treatment of Glaucoma, Age Related Macular Degeneration and Uveitis.Recent Pat Biomed Eng. 2012 Apr 1;5(1):83-101. doi: 10.2174/1874764711205010083. Recent Pat Biomed Eng. 2012. PMID: 25414810 Free PMC article.
-
Medical Management of Glaucoma in the 21st Century from a Canadian Perspective.J Ophthalmol. 2016;2016:6509809. doi: 10.1155/2016/6509809. Epub 2016 Nov 8. J Ophthalmol. 2016. PMID: 27895937 Free PMC article. Review.
-
Novel ocular antihypertensive compounds in clinical trials.Clin Ophthalmol. 2011;5:667-77. doi: 10.2147/OPTH.S15971. Epub 2011 May 20. Clin Ophthalmol. 2011. PMID: 21629573 Free PMC article.
-
Regulatory Roles of Anoctamin-6 in Human Trabecular Meshwork Cells.Invest Ophthalmol Vis Sci. 2017 Jan 1;58(1):492-501. doi: 10.1167/iovs.16-20188. Invest Ophthalmol Vis Sci. 2017. PMID: 28125837 Free PMC article.
-
Nucleoside-derived antagonists to A3 adenosine receptors lower mouse intraocular pressure and act across species.Exp Eye Res. 2010 Jan;90(1):146-54. doi: 10.1016/j.exer.2009.10.001. Epub 2009 Oct 28. Exp Eye Res. 2010. PMID: 19878673 Free PMC article.
References
-
- Al-Aswad LA, Gong H, Lee D, O’Donnell ME, Brandt JD, Ryan WJ, Schroeder A, Erickson KA. Effects of Na-K-2Cl cotransport regulators on outflow facility in calf and human eyes in vitro. Invest Ophthalmol Vis Sci. 1999;40:1695–1701. - PubMed
-
- Avila MY, Carré DA, Stone RA, Civan MM. Reliable measurement of mouse intraocular pressure by a servo-null micropipette system. Invest Ophthalmol Vis Sci. 2001a;42:1841–1846. - PubMed
-
- Avila MY, Seidler RW, Stone RA, Civan MM. Inhibitors of NHE-1 Na+/H+ exchange reduce mouse intraocular pressure. Invest Ophthalmol Vis Sci. 2002a;43:1897–1902. - PubMed
-
- Avila MY, Stone RA, Civan MM. Knockout of A(3) adenosine receptors reduces mouse intraocular pressure. Invest Ophthalmol Vis Sci. 2002b;43:3021–3026. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

