Rofecoxib modulates multiple gene expression pathways in a clinical model of acute inflammatory pain
- PMID: 17070997
- PMCID: PMC1894940
- DOI: 10.1016/j.pain.2006.09.011
Rofecoxib modulates multiple gene expression pathways in a clinical model of acute inflammatory pain
Abstract
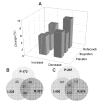
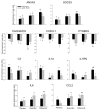
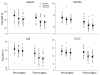
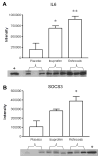
New insights into the biological properties of cyclooxygenase-2 (COX-2) and its response pathway challenge the hypothesis that COX-2 is simply pro-inflammatory and inhibition of COX-2 solely prevents the development of inflammation and ameliorates inflammatory pain. The present study performed a comprehensive analysis of gene/protein expression induced by a selective inhibitor of COX-2, rofecoxib, compared with a non-selective COX inhibitor, ibuprofen, and placebo in a clinical model of acute inflammatory pain (the surgical extraction of impacted third molars) using microarray analysis followed by quantitative RT-PCR verification and Western blotting. Inhibition of COX-2 modulated gene expression related to inflammation and pain, the arachidonic acid pathway, apoptosis/angiogenesis, cell adhesion and signal transduction. Compared to placebo, rofecoxib treatment increased the gene expression of ANXA3 (annexin 3), SOD2 (superoxide dismutase 2), SOCS3 (suppressor of cytokine signaling 3) and IL1RN (IL1 receptor antagonist) which are associated with inhibition of phospholipase A(2) and suppression of cytokine signaling cascades, respectively. Both rofecoxib and ibuprofen treatment increased the gene expression of the pro-inflammatory mediators, IL6 and CCL2 (chemokine C-C motif ligand 2), following tissue injury compared to the placebo treatment. These results indicate a complex role for COX-2 in the inflammatory cascade in addition to the well-characterized COX-dependent pathway, as multiple pathways are also involved in rofecoxib-induced anti-inflammatory and analgesic effects at the gene expression level. These findings may also suggest an alternative hypothesis for the adverse effects attributed to selective inhibition of COX-2.
Conflict of interest statement
Figures
Similar articles
-
Rofecoxib regulates the expression of genes related to the matrix metalloproteinase pathway in humans: implication for the adverse effects of cyclooxygenase-2 inhibitors.Clin Pharmacol Ther. 2006 Apr;79(4):303-15. doi: 10.1016/j.clpt.2005.12.306. Clin Pharmacol Ther. 2006. PMID: 16580899 Clinical Trial.
-
Ibuprofen as a pre-emptive analgesic is as effective as rofecoxib for mandibular third molar surgery.Odontology. 2006 Sep;94(1):59-63. doi: 10.1007/s10266-006-0062-4. Odontology. 2006. PMID: 16998619 Clinical Trial.
-
Efficacy of preoperative oral rofecoxib in pain control for third molar surgery.Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005 Jun;99(6):e47-53. doi: 10.1016/j.tripleo.2005.02.075. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005. PMID: 15897845 Clinical Trial.
-
Selective COX-2 inhibition and cardiovascular effects: a review of the rofecoxib development program.Am Heart J. 2003 Oct;146(4):591-604. doi: 10.1016/S0002-8703(03)00398-3. Am Heart J. 2003. PMID: 14564311 Review.
-
Relative efficacy of selective COX-2 inhibitors compared with over-the-counter ibuprofen.Int J Clin Pract Suppl. 2003 Apr;(135):18-22. Int J Clin Pract Suppl. 2003. PMID: 12723742 Review.
Cited by
-
Cytokine patterns in brain tumour progression.Mediators Inflamm. 2013;2013:979748. doi: 10.1155/2013/979748. Epub 2013 Jun 24. Mediators Inflamm. 2013. PMID: 23864770 Free PMC article.
-
Genomic Profile of Fatigued Men Receiving Localized Radiation Therapy.Biol Res Nurs. 2016 May;18(3):281-9. doi: 10.1177/1099800415618786. Epub 2015 Nov 29. Biol Res Nurs. 2016. PMID: 26620220 Free PMC article.
-
Genomics of pain in osteoarthritis.Osteoarthritis Cartilage. 2013 Sep;21(9):1374-82. doi: 10.1016/j.joca.2013.06.010. Osteoarthritis Cartilage. 2013. PMID: 23973152 Free PMC article. Review.
-
Upregulation of IL-6, IL-8 and CCL2 gene expression after acute inflammation: Correlation to clinical pain.Pain. 2009 Apr;142(3):275-283. doi: 10.1016/j.pain.2009.02.001. Epub 2009 Feb 23. Pain. 2009. PMID: 19233564 Free PMC article.
-
Genomic methods for clinical and translational pain research.Methods Mol Biol. 2012;851:9-46. doi: 10.1007/978-1-61779-561-9_2. Methods Mol Biol. 2012. PMID: 22351080 Free PMC article.
References
-
- Alexander WS. Suppressors of cytokine signaling (SOCS) in the immune system. Nat Rev Immunol. 2002;2:410–16. - PubMed
-
- Alexander WS, Hilton DJ. The role of suppressors of cytokine signaling (SOCS) proteins in regulation of the immune response. Annu Rev Immunol. 2004;22:503–29. - PubMed
-
- Ben-Av P, Crofford LJ, Wilder RL, Hla T. Induction of vascular endothelial growth factor expression in synovial fibroblasts by prostaglandin E and interleukin-1: a potential mechanism for inflammatory angiogenesis. FEBS Lett. 1995;372:83–7. - PubMed
-
- Bingham S, Beswick PJ, Bountra C, Brown T, Campbell IB, Chessell IP, Clayton N, Collins SD, Davey PT, Goodland H, Gray N, Haslam C, Hatcher JP, Hunter AJ, Lucas F, Murkitt G, Naylor A, Pickup E, Sargent B, Summerfield SG, Stevens A, Stratton SC, Wiseman J. The cyclooxygenase-2 inhibitor GW406381X [2-(4-ethoxyphenyl)-3-[4-(methylsulfonyl)phenyl]-pyrazolo[1,5-b]pyridazine] is effective in animal models of neuropathic pain and central sensitization. J Pharmacol Exp Ther. 2005;312:1161–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

