Vasopressin stimulates action potential firing by protein kinase C-dependent inhibition of KCNQ5 in A7r5 rat aortic smooth muscle cells
- PMID: 17071736
- PMCID: PMC2577603
- DOI: 10.1152/ajpheart.00065.2006
Vasopressin stimulates action potential firing by protein kinase C-dependent inhibition of KCNQ5 in A7r5 rat aortic smooth muscle cells
Abstract
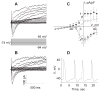
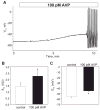
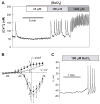
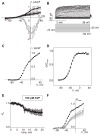
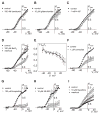
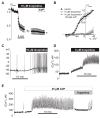
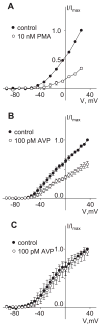
[Arg(8)]-vasopressin (AVP), at low concentrations (10-500 pM), stimulates oscillations in intracellular Ca(2+) concentration (Ca(2+) spikes) in A7r5 rat aortic smooth muscle cells. Our previous studies provided biochemical evidence that protein kinase C (PKC) activation and phosphorylation of voltage-sensitive K(+) (K(v)) channels are crucial steps in this process. In the present study, K(v) currents (I(Kv)) and membrane potential were measured using patch clamp techniques. Treatment of A7r5 cells with 100 pM AVP resulted in significant inhibition of I(Kv). This effect was associated with gradual membrane depolarization, increased membrane resistance, and action potential (AP) generation in the same cells. The AVP-sensitive I(Kv) was resistant to 4-aminopyridine, iberiotoxin, and glibenclamide but was fully inhibited by the selective KCNQ channel blockers linopirdine (10 microM) and XE-991 (10 microM) and enhanced by the KCNQ channel activator flupirtine (10 microM). BaCl(2) (100 microM) or linopirdine (5 microM) mimicked the effects of AVP on K(+) currents, AP generation, and Ca(2+) spiking. Expression of KCNQ5 was detected by RT-PCR in A7r5 cells and freshly isolated rat aortic smooth muscle. RNA interference directed toward KCNQ5 reduced KCNQ5 protein expression and resulted in a significant decrease in I(Kv) in A7r5 cells. I(Kv) was also inhibited in response to the PKC activator 4beta-phorbol 12-myristate 13-acetate (10 nM), and the inhibition of I(Kv) by AVP was prevented by the PKC inhibitor calphostin C (250 nM). These results suggest that the stimulation of Ca(2+) spiking by physiological concentrations of AVP involves PKC-dependent inhibition of KCNQ5 channels and increased AP firing in A7r5 cells.
Figures


References
-
- Byron KL, Lucchesi PA. Signal transduction of physiological concentrations of vasopressin in A7r5 vascular smooth muscle cells. J Biol Chem. 2002;277:7298–7307. - PubMed
-
- Byron KL, Villereal ML. Mitogen-induced [Ca2+]i changes in individual human fibroblasts. Image analysis reveals asynchronous responses which are characteristic for different mitogens. J Biol Chem. 1989;264:18234–18239. - PubMed
-
- Byron KL. Vasopressin stimulates Ca2+ spiking activity in A7r5 vascular smooth muscle cells via activation of phospholipase A2. Circ Res. 1996;78:813–820. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

