Two separate molecular systems, Dachsous/Fat and Starry night/Frizzled, act independently to confer planar cell polarity
- PMID: 17075008
- PMCID: PMC2747022
- DOI: 10.1242/dev.02641
Two separate molecular systems, Dachsous/Fat and Starry night/Frizzled, act independently to confer planar cell polarity
Abstract
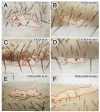
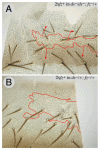
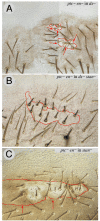
Planar polarity is a fundamental property of epithelia in animals and plants. In Drosophila it depends on at least two sets of genes: one set, the Ds system, encodes the cadherins Dachsous (Ds) and Fat (Ft), as well as the Golgi protein Four-jointed. The other set, the Stan system, encodes Starry night (Stan or Flamingo) and Frizzled. The prevailing view is that the Ds system acts via the Stan system to orient cells. However, using the Drosophila abdomen, we find instead that the two systems operate independently: each confers and propagates polarity, and can do so in the absence of the other. We ask how the Ds system acts; we find that either Ds or Ft is required in cells that send information and we show that both Ds and Ft are required in the responding cells. We consider how polarity may be propagated by Ds-Ft heterodimers acting as bridges between cells.
Figures




References
-
- Adler PN. Planar signaling and morphogenesis in Drosophila. Dev Cell. 2002;2:525–35. - PubMed
-
- Adler PN, Charlton J, Liu J. Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype by altering frizzled signaling. Development. 1998;125:959–68. - PubMed
-
- Adler PN, Krasnow RE, Liu J. Tissue polarity points from cells that have higher Frizzled levels towards cells that have lower Frizzled levels. Curr Biol. 1997;7:940–9. - PubMed
-
- Amonlirdviman K, Khare NA, Tree DR, Chen WS, Axelrod JD, Tomlin CJ. Mathematical modeling of planar cell polarity to understand domineering nonautonomy. Science. 2005;307:423–6. - PubMed
-
- Baena-López LA, Baonza A, Garcia-Bellido A. The orientation of cell divisions determines the shape of Drosophila organs. Curr Biol. 2005;15:1640–4. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

