Fusion of microglia with pyramidal neurons after retroviral infection
- PMID: 17079670
- PMCID: PMC6674527
- DOI: 10.1523/JNEUROSCI.3340-06.2006
Fusion of microglia with pyramidal neurons after retroviral infection
Abstract
The neurogenic potential of the postnatal neocortex has not been tested previously with a combination of both retroviral and bromodeoxyuridine (BrdU) labeling. Here we report that injections of enhanced green fluorescent protein (eGFP) retrovirus into 134 postnatal rats resulted in GFP labeling of 642 pyramidal neurons in neocortex. GFP-labeled neocortical pyramidal neurons, however, unlike GFP-labeled glia, did not incorporate BrdU. Closer inspection of retrovirally labeled neurons revealed microglia fused to the apical dendrites of labeled pyramidal neurons. Retroviral infection of mixed cultures of cortical neurons and glia confirmed the presence of specific neuronal-microglial fusions. Microglia did not fuse to other glial cell types, and cultures not treated with retrovirus lacked microglial-neuronal fusion. Furthermore, activation of microglia by lipopolysaccharide greatly increased the virally induced fusion of microglia to neurons in culture. These results indicate a novel form of specific cell fusion between neuronal dendrites and microglia and further illustrate the need for caution when interpreting evidence for neuronogenesis in the postnatal brain.
Figures
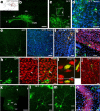
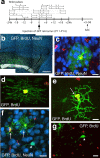
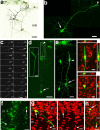
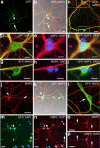
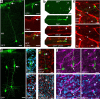
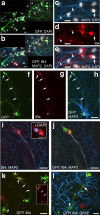
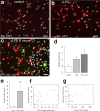
Comment in
-
Glowing green pyramids: a false positive for neocortical neurogenesis reveals a novel neuronal-microglial fusion in the postnatal brain.J Neurosci. 2007 Feb 14;27(7):1507-8. doi: 10.1523/jneurosci.5475-06.2007. J Neurosci. 2007. PMID: 17304702 Free PMC article. Review. No abstract available.
References
-
- Alvarez-Dolado M, Pardal R, Garcia-Verdugo JM, Fike JR, Lee HO, Pfeffer K, Lois C, Morrison SJ, Alvarez-Buylla A. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature. 2003;425:968–973. - PubMed
-
- Anderson JM. Multinucleated giant cells. Curr Opin Hematol. 2000;7:40–47. - PubMed
-
- Blumenthal R, Bali-Puri A, Walter A, Covell D, Eidelman O. pH-dependent fusion of vesicular stomatitis virus with Vero cells. Measurement by dequenching of octadecyl rhodamine fluorescence. J Biol Chem. 1987;262:13614–13619. - PubMed
-
- Brazelton TR, Rossi FM, Keshet GI, Blau HM. From marrow to brain: expression of neuronal phenotypes in adult mice. Science. 2000;290:1775–1779. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
