A postsynaptic spectrin scaffold defines active zone size, spacing, and efficacy at the Drosophila neuromuscular junction
- PMID: 17088429
- PMCID: PMC2064525
- DOI: 10.1083/jcb.200607036
A postsynaptic spectrin scaffold defines active zone size, spacing, and efficacy at the Drosophila neuromuscular junction
Abstract
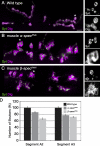
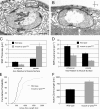
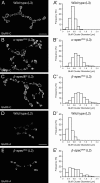
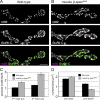
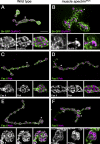
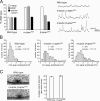
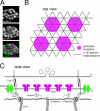
Synaptic connections are established with characteristic, cell type-specific size and spacing. In this study, we document a role for the postsynaptic Spectrin skeleton in this process. We use transgenic double-stranded RNA to selectively eliminate alpha-Spectrin, beta-Spectrin, or Ankyrin. In the absence of postsynaptic alpha- or beta-Spectrin, active zone size is increased and spacing is perturbed. In addition, subsynaptic muscle membranes are significantly altered. However, despite these changes, the subdivision of the synapse into active zone and periactive zone domains remains intact, both pre- and postsynaptically. Functionally, altered active zone dimensions correlate with an increase in quantal size without a change in presynaptic vesicle size. Mechanistically, beta-Spectrin is required for the localization of alpha-Spectrin and Ankyrin to the postsynaptic membrane. Although Ankyrin is not required for the localization of the Spectrin skeleton to the neuromuscular junction, it contributes to Spectrin-mediated synapse development. We propose a model in which a postsynaptic Spectrin-actin lattice acts as an organizing scaffold upon which pre- and postsynaptic development are arranged.
Figures


Similar articles
-
The spectrin-based skeleton at the postsynaptic membrane of the neuromuscular junction.Microsc Res Tech. 2000 Apr 1;49(1):101-7. doi: 10.1002/(SICI)1097-0029(20000401)49:1<101::AID-JEMT11>3.0.CO;2-U. Microsc Res Tech. 2000. PMID: 10757883 Review.
-
Postsynaptic actin regulates active zone spacing and glutamate receptor apposition at the Drosophila neuromuscular junction.Mol Cell Neurosci. 2014 Jul;61:241-54. doi: 10.1016/j.mcn.2014.07.005. Epub 2014 Jul 24. Mol Cell Neurosci. 2014. PMID: 25066865 Free PMC article.
-
Tenectin recruits integrin to stabilize bouton architecture and regulate vesicle release at the Drosophila neuromuscular junction.Elife. 2018 Jun 14;7:e35518. doi: 10.7554/eLife.35518. Elife. 2018. PMID: 29901439 Free PMC article.
-
Morphology and molecular organization of the adult neuromuscular junction of Drosophila.J Comp Neurol. 2004 Jan 19;468(4):596-613. doi: 10.1002/cne.10977. J Comp Neurol. 2004. PMID: 14689489
-
Synaptic homeostasis on the fast track.Neuron. 2006 Nov 22;52(4):569-71. doi: 10.1016/j.neuron.2006.11.006. Neuron. 2006. PMID: 17114040 Review.
Cited by
-
Developmental arrest of Drosophila larvae elicits presynaptic depression and enables prolonged studies of neurodegeneration.Development. 2020 May 21;147(10):dev186312. doi: 10.1242/dev.186312. Development. 2020. PMID: 32345746 Free PMC article.
-
Syndapin promotes formation of a postsynaptic membrane system in Drosophila.Mol Biol Cell. 2009 Apr;20(8):2254-64. doi: 10.1091/mbc.e08-10-1072. Epub 2009 Feb 25. Mol Biol Cell. 2009. PMID: 19244343 Free PMC article.
-
Nervous wreck interacts with thickveins and the endocytic machinery to attenuate retrograde BMP signaling during synaptic growth.Neuron. 2008 May 22;58(4):507-18. doi: 10.1016/j.neuron.2008.03.007. Neuron. 2008. PMID: 18498733 Free PMC article.
-
Active zones of mammalian neuromuscular junctions: formation, density, and aging.Ann N Y Acad Sci. 2012 Dec;1274(1):24-32. doi: 10.1111/j.1749-6632.2012.06836.x. Ann N Y Acad Sci. 2012. PMID: 23252894 Free PMC article. Review.
-
Vgsc-interacting proteins are genetically associated with pyrethroid resistance in Aedes aegypti.PLoS One. 2019 Jan 29;14(1):e0211497. doi: 10.1371/journal.pone.0211497. eCollection 2019. PLoS One. 2019. PMID: 30695054 Free PMC article.
References
-
- Ango, F., G. di Cristo, H. Higashiyama, V. Bennett, P. Wu, and Z.J. Huang. 2004. Ankyrin-based subcellular gradient of neurofascin, an immunoglobulin family protein, directs GABAergic innervation at Purkinje axon initial segment. Cell. 119:257–272. - PubMed
-
- Atwood, H.L., C.K. Govind, and C.F. Wu. 1993. Differential ultrastructure of synaptic terminals on ventral longitudinal abdominal muscles in Drosophila larvae. J. Neurobiol. 24:1008–1024. - PubMed
-
- Bennett, V., and A.J. Baines. 2001. Spectrin and ankyrin-based pathways: metazoan inventions for integrating cells into tissues. Physiol. Rev. 81:1353–1392. - PubMed
-
- Bennett, V., J. Davis, and W.E. Fowler. 1982. Brain spectrin, a membrane-associated protein related in structure and function to erythrocyte spectrin. Nature. 299:126–131. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

