A functional model for the cysteinate-ligated non-heme iron enzyme superoxide reductase (SOR)
- PMID: 17090014
- PMCID: PMC2532059
- DOI: 10.1021/ja064870d
A functional model for the cysteinate-ligated non-heme iron enzyme superoxide reductase (SOR)
Abstract
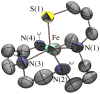
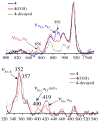
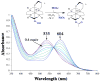
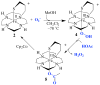
Superoxide reductases (SORs) are cysteine-ligated, non-heme iron enzymes that reduce toxic superoxide radicals (O2-). The functional role of the trans cysteinate, as well as the mechanism by which SOR reduces O2-, is unknown. Herein is described a rare example of a functional metalloenzyme analogue, which catalytically reduces superoxide in a proton-dependent mechanism, via a trans thiolate-ligated iron-peroxo intermediate, the first example of its type. Acetic-acid-promoted H2O2 release, followed by Cp2Co reduction, regenerates the active Fe(II) catalyst. The thiolate ligand and its trans positioning relative to the substrate are shown to contribute significantly to the catalyst's function, by lowering the redox potential, changing the spin state, and dramatically lowering the nuFe-O stretching frequency well-below that of any other reported iron-peroxo, while leaving nuO-O high, so as to favor superoxide reduction and Fe-O, as opposed to O-O, bond cleavage. Thus we provide critical insight into the relationship between the SOR structure and its function, as well as important benchmark parameters for characterizing highly unstable thiolate-ligated iron-peroxo intermediates.
Figures
Similar articles
-
Influence of thiolate ligands on reductive N-O bond activation. Probing the O2(-) binding site of a biomimetic superoxide reductase analogue and examining the proton-dependent reduction of nitrite.J Am Chem Soc. 2011 Feb 9;133(5):1419-27. doi: 10.1021/ja107551u. Epub 2011 Jan 5. J Am Chem Soc. 2011. PMID: 21207999 Free PMC article.
-
Understanding how the thiolate sulfur contributes to the function of the non-heme iron enzyme superoxide reductase.Acc Chem Res. 2007 Jul;40(7):501-9. doi: 10.1021/ar600059h. Epub 2007 May 31. Acc Chem Res. 2007. PMID: 17536780 Free PMC article. Review.
-
Fe(3+)-eta(2)-peroxo species in superoxide reductase from Treponema pallidum. Comparison with Desulfoarculus baarsii.Biophys Chem. 2006 Jan 1;119(1):38-48. doi: 10.1016/j.bpc.2005.06.013. Epub 2005 Aug 9. Biophys Chem. 2006. PMID: 16084640
-
Conformational H-bonding modulation of the iron active site cysteine ligand of superoxide reductase: absorption and resonance Raman studies.Phys Chem Chem Phys. 2021 Feb 28;23(8):4636-4645. doi: 10.1039/d0cp03898a. Epub 2021 Feb 2. Phys Chem Chem Phys. 2021. PMID: 33527107
-
Binding of cysteine thiolate to the Fe(III) heme complex is critical for the function of heme sensor proteins.J Inorg Biochem. 2012 Mar;108:171-7. doi: 10.1016/j.jinorgbio.2011.08.018. Epub 2011 Aug 28. J Inorg Biochem. 2012. PMID: 22137591 Review.
Cited by
-
Influence of thiolate ligands on reductive N-O bond activation. Probing the O2(-) binding site of a biomimetic superoxide reductase analogue and examining the proton-dependent reduction of nitrite.J Am Chem Soc. 2011 Feb 9;133(5):1419-27. doi: 10.1021/ja107551u. Epub 2011 Jan 5. J Am Chem Soc. 2011. PMID: 21207999 Free PMC article.
-
Addition of dioxygen to an N4S(thiolate) iron(II) cysteine dioxygenase model gives a structurally characterized sulfinato-iron(II) complex.J Am Chem Soc. 2012 May 30;134(21):8758-61. doi: 10.1021/ja302112y. Epub 2012 May 17. J Am Chem Soc. 2012. PMID: 22578255 Free PMC article.
-
How Do Ring Size and π-Donating Thiolate Ligands Affect Redox-Active, α-Imino-N-heterocycle Ligand Activation?Inorg Chem. 2018 Feb 19;57(4):1935-1949. doi: 10.1021/acs.inorgchem.7b02748. Epub 2018 Feb 7. Inorg Chem. 2018. PMID: 29411979 Free PMC article.
-
Leveraging ligand-based proton and electron transfer for aerobic reactivity and catalysis.Chem Sci. 2024 Sep 9;15(40):16409-23. doi: 10.1039/d4sc03896g. Online ahead of print. Chem Sci. 2024. PMID: 39386904 Free PMC article. Review.
-
Correlation between structural, spectroscopic, and reactivity properties within a series of structurally analogous metastable manganese(III)-alkylperoxo complexes.J Am Chem Soc. 2013 Mar 20;135(11):4260-72. doi: 10.1021/ja308915x. Epub 2013 Mar 12. J Am Chem Soc. 2013. PMID: 23432090 Free PMC article.
References
-
- Kurtz DM., Jr Acc Chem Res. 2004;37:902–908. - PubMed
- Mathé C, Nivire V, Houée-Levin C, Mattioli TA. Biophys Chem. 2006;119:38–48. - PubMed
- Yeh AP, Hu Y, Jenney FE, Jr, Adams MWW, Rees DC. Biochemistry. 2000;39:2499–2508. - PubMed
- Emerson JP, Coulter ED, Cabelli DE, Phillips RS, Kurtz DM., Jr Biochemistry. 2002;41:4348–4357. - PubMed
- Niviere V, Asso M, Weill CO, Lombard M, et al. Biochemistry. 2004;43:808–818. - PubMed
- Mathe C, Mattioli TA, Horner O, Lombard M, Latour J-M, Fontecave M, Niviere V. J Am Chem Soc. 2002;124:4966–4967. - PubMed
- Horner O, Oddou J–L, Niviere V, Fontecave M, Halfen JA, Latour J-M, et al. Biochemistry. 2004;43:8815–8825. - PubMed
- Mathé C, Nivière V, Mattioli TA. J Am Chem Soc. 2005;127:16436–16441. - PubMed
- Clay MD, Jenney FE, Jr, Hagedoorn PL, George GN, Adams MWW, Johnson MK. J Am Chem Soc. 2002;124:788–805. - PubMed
- Jovanovic T, Krebs C, Moura I, Moura JJG, Radolf JD, Huynh BH, Rusnak F, et al. J Biol Chem. 2000;275:28439–28448. - PubMed
-
- Gogun Y, Sakurada S, Kimura Y, Nagumo M. J Clin Biochem Nutr. 1990;8:85–92.
- Kocaturk PA, Akbostanci MC, Tan F, Kavas GO. Pathophysiology. 2000;7:63–67. - PubMed
- Ihara Y, Chuda M, Kuroda S, Hayabara T. J Neurol Sci. 1999;170:90–95. - PubMed
- De Leo ME, Borrello S, Passantino M, Palazzotti B, Galeotti T, Masullo C, et al. Neurosci Lett. 1998;250:173–176. - PubMed
-
- Addision AW, Rao TN, Reedijk J. J Chem Soc Dalton Trans. 1984:1349.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

