The role of nicotinic acetylcholine receptors in the primary reinforcing and reinforcement-enhancing effects of nicotine
- PMID: 17091131
- PMCID: PMC2907080
- DOI: 10.1038/sj.npp.1301228
The role of nicotinic acetylcholine receptors in the primary reinforcing and reinforcement-enhancing effects of nicotine
Abstract
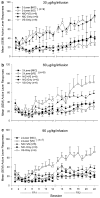
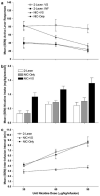
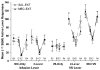
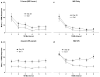
The primary reinforcing effects of nicotine are mediated by the drugs action at central nervous system nicotinic acetylcholine receptors (nAChRs). Although previous studies have demonstrated that nicotine potently enhances responding for non-pharmacological stimuli, the role of nAChRs in this reinforcement-enhancing effect is not known. The two reinforcement-related effects of nicotine can be dissociated in a paradigm that provides concurrent access to drug infusions and a non-pharmacological visual stimulus (VS). The present study characterized the role of nAChRs in the primary reinforcing effect of nicotine and the reinforcement-enhancing effect of nicotine. For rats with access to VS (VS-Only), nicotine (NIC-Only), both reinforcers contingent upon one response (NIC+VS) or both reinforcers contingent upon separate responses (2-Lever), unit dose-response relationships (0, 30, 60, or 90 microg/kg/infusion, free base) were determined over a 22-day acquisition period. Expression of the two reinforcement-related effects of nicotine was manipulated by pharmacological antagonism of nAChRs (1 mg/kg mecamylamine, subcutaneous, 5-min before the session) or by substituting saline for nicotine infusions (ie extinction) over a series of seven test sessions. Unit dose manipulations yielded an inverse dose-response relationship for active lever responding in the NIC+VS group. The dose-response relationships for rats with independent access to each reinforcer (2-Lever group) were relatively flat. For the 2-Lever group, acute mecamylamine challenge blocked the reinforcement-enhancing effects of nicotine, VS-lever responding decreased to basal levels on the first day of mecamylamine treatment or saline substitution (to the level of the VS-Only group). In contrast, nicotine-lever responding decreased gradually over the 7-day testing period (similar to saline extinction). The two reinforcement-related effects of nicotine are mediated by nAChRs but can be dissociated by acute and chronic profiles.
Figures
Similar articles
-
Reinforcement enhancing effect of nicotine and its attenuation by nicotinic antagonists in rats.Psychopharmacology (Berl). 2007 Nov;194(4):463-73. doi: 10.1007/s00213-007-0863-3. Epub 2007 Jul 8. Psychopharmacology (Berl). 2007. PMID: 17616849 Free PMC article.
-
Neurochemical and behavioral effects of bupropion and mecamylamine in the presence of nicotine.Brain Res. 2006 Oct 30;1117(1):18-24. doi: 10.1016/j.brainres.2006.07.110. Epub 2006 Sep 1. Brain Res. 2006. PMID: 16949560
-
Bupropion and nicotine enhance responding for nondrug reinforcers via dissociable pharmacological mechanisms in rats.Psychopharmacology (Berl). 2009 Dec;207(3):381-90. doi: 10.1007/s00213-009-1666-5. Epub 2009 Sep 17. Psychopharmacology (Berl). 2009. PMID: 19760281 Free PMC article.
-
The neuropharmacological substrates of nicotine reward: reinforcing versus reinforcement-enhancing effects of nicotine.Behav Pharmacol. 2009 May;20(3):211-25. doi: 10.1097/FBP.0b013e32832c7083. Behav Pharmacol. 2009. PMID: 19421028 Review.
-
It is not "either/or": activation and desensitization of nicotinic acetylcholine receptors both contribute to behaviors related to nicotine addiction and mood.Prog Neurobiol. 2008 Apr;84(4):329-42. doi: 10.1016/j.pneurobio.2007.12.005. Epub 2007 Dec 27. Prog Neurobiol. 2008. PMID: 18242816 Free PMC article. Review.
Cited by
-
The effect of nicotine on sign-tracking and goal-tracking in a Pavlovian conditioned approach paradigm in rats.Psychopharmacology (Berl). 2013 Mar;226(2):247-59. doi: 10.1007/s00213-012-2892-9. Epub 2012 Oct 23. Psychopharmacology (Berl). 2013. PMID: 23090624
-
Adolescent Rats Self-Administer Less Nicotine Than Adults at Low Doses.Nicotine Tob Res. 2016 Sep;18(9):1861-1868. doi: 10.1093/ntr/ntw006. Epub 2016 Jan 13. Nicotine Tob Res. 2016. PMID: 26764255 Free PMC article.
-
Expression of sensitized β2 nAChR subunits in VTA neurons enhances intravenous nicotine self-administration in male rats.Neuropharmacology. 2024 Dec 15;261:110161. doi: 10.1016/j.neuropharm.2024.110161. Epub 2024 Sep 17. Neuropharmacology. 2024. PMID: 39299573
-
Conditioned reinforcement in rats established with self-administered nicotine and enhanced by noncontingent nicotine.Psychopharmacology (Berl). 2007 Dec;195(2):235-43. doi: 10.1007/s00213-007-0897-6. Epub 2007 Aug 5. Psychopharmacology (Berl). 2007. PMID: 17676401 Free PMC article.
-
Lack of evidence for positive reinforcing and prosocial effects of MDMA in pair-housed male and female rats.Eur J Pharmacol. 2021 Dec 15;913:174646. doi: 10.1016/j.ejphar.2021.174646. Epub 2021 Nov 17. Eur J Pharmacol. 2021. PMID: 34800468 Free PMC article.
References
-
- Balfour DJ, Wright AE, Benwell ME, Birrell CE. The putative role of extra-synaptic mesolimbic dopamine in the neurobiology of nicotine dependence. Behav Brain Res. 2000;113:73–83. - PubMed
-
- Bevins RA, Palmatier MI. Effects of nicotine preexposure on the conditioned and unconditioned psychomotor effects of nicotine in rats. Behav Brain Res. 2003;143:65–74. - PubMed
-
- Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, et al. Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology. 2002;163:230–237. - PubMed
-
- Chaudhri N, Caggiula AR, Donny EC, Booth S, Gharib M, Craven L, et al. Dissociating the dual-actions of nicotine: primary reinforcement and the reinforcement-enhancing actions of nicotine impact operant responding for a visual stimulus in rats. Psychopharmacology. 2006c doi:10.1007/s00213-006-0454-8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

