Endocytic recycling in yeast is regulated by putative phospholipid translocases and the Ypt31p/32p-Rcy1p pathway
- PMID: 17093059
- PMCID: PMC1751321
- DOI: 10.1091/mbc.e06-05-0461
Endocytic recycling in yeast is regulated by putative phospholipid translocases and the Ypt31p/32p-Rcy1p pathway
Abstract
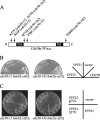
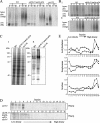
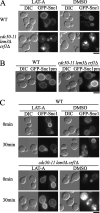
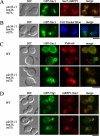
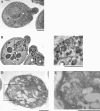
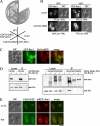
Phospholipid translocases (PLTs) have been implicated in the generation of phospholipid asymmetry in membrane bilayers. In budding yeast, putative PLTs are encoded by the DRS2 gene family of type 4 P-type ATPases. The homologous proteins Cdc50p, Lem3p, and Crf1p are potential noncatalytic subunits of Drs2p, Dnf1p and Dnf2p, and Dnf3p, respectively; these putative heteromeric PLTs share an essential function for cell growth. We constructed temperature-sensitive mutants of CDC50 in the lem3Delta crf1Delta background (cdc50-ts mutants). Screening for multicopy suppressors of cdc50-ts identified YPT31/32, two genes that encode Rab family small GTPases that are involved in both the exocytic and endocytic recycling pathways. The cdc50-ts mutants did not exhibit major defects in the exocytic pathways, but they did exhibit those in endocytic recycling; large membranous structures containing the vesicle-soluble N-ethylmaleimide-sensitive factor attachment protein receptor Snc1p intracellularly accumulated in these mutants. Genetic results suggested that the YPT31/32 effector RCY1 and CDC50 function in the same signaling pathway, and simultaneous overexpression of CDC50, DRS2, and GFP-SNC1 restored growth as well as the plasma membrane localization of GFP-Snc1p in the rcy1Delta mutant. In addition, Rcy1p coimmunoprecipitated with Cdc50p-Drs2p. We propose that the Ypt31p/32p-Rcy1p pathway regulates putative phospholipid translocases to promote formation of vesicles destined for the trans-Golgi network from early endosomes.
Figures



References
-
- Bai C., Sen P., Hofmann K., Ma L., Goebl M., Harper J. W., Elledge S. J. SKP1 connects cell cycle regulators to the ubiquitin proteolysis machinery through a novel motif, the F-box. Cell. 1996;86:263–274. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

