The yeast PH domain proteins Slm1 and Slm2 are targets of sphingolipid signaling during the response to heat stress
- PMID: 17101780
- PMCID: PMC1800798
- DOI: 10.1128/MCB.00461-06
The yeast PH domain proteins Slm1 and Slm2 are targets of sphingolipid signaling during the response to heat stress
Abstract
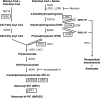
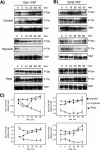
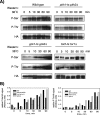
The PH domain-containing proteins Slm1 and Slm2 were previously identified as effectors of the phosphatidylinositol-4,5-bisphosphate (PI4,5P(2)) and TORC2 signaling pathways. Here, we demonstrate that Slm1 and Slm2 are also targets of sphingolipid signaling during the heat shock response. We show that upon depletion of cellular sphingolipid levels, Slm1 function becomes essential for survival under heat stress. We further demonstrate that Slm proteins are regulated by a phosphorylation/dephosphorylation cycle involving the sphingolipid-activated protein kinases Pkh1 and Pkh2 and the calcium/calmodulin-dependent protein phosphatase calcineurin. By using a combination of mass spectrometry and mutational analysis, we identified serine residue 659 in Slm1 as a site of phosphorylation. Characterization of Slm1 mutants that mimic dephosphorylated and phosphorylated states demonstrated that phosphorylation at serine 659 is vital for survival under heat stress and promotes the proper polarization of the actin cytoskeleton. Finally, we present evidence that Slm proteins are also required for the trafficking of the raft-associated arginine permease Can1 to the plasma membrane, a process that requires sphingolipid synthesis and actin polymerization. Together with previous work, our findings suggest that Slm proteins are subject to regulation by multiple signals, including PI4,5P(2), TORC2, and sphingolipids, and may thus integrate inputs from different signaling pathways to temporally and spatially control actin polarization.
Figures







Similar articles
-
Slm1 and slm2 are novel substrates of the calcineurin phosphatase required for heat stress-induced endocytosis of the yeast uracil permease.Mol Cell Biol. 2006 Jun;26(12):4729-45. doi: 10.1128/MCB.01973-05. Mol Cell Biol. 2006. PMID: 16738335 Free PMC article.
-
The pleckstrin homology domain proteins Slm1 and Slm2 are required for actin cytoskeleton organization in yeast and bind phosphatidylinositol-4,5-bisphosphate and TORC2.Mol Biol Cell. 2005 Apr;16(4):1883-900. doi: 10.1091/mbc.e04-07-0564. Epub 2005 Feb 2. Mol Biol Cell. 2005. PMID: 15689497 Free PMC article.
-
The phosphatidylinositol 4,5-biphosphate and TORC2 binding proteins Slm1 and Slm2 function in sphingolipid regulation.Mol Cell Biol. 2006 Aug;26(15):5861-75. doi: 10.1128/MCB.02403-05. Mol Cell Biol. 2006. PMID: 16847337 Free PMC article.
-
Thematic review series: sphingolipids. New insights into sphingolipid metabolism and function in budding yeast.J Lipid Res. 2008 May;49(5):909-21. doi: 10.1194/jlr.R800003-JLR200. Epub 2008 Feb 23. J Lipid Res. 2008. PMID: 18296751 Free PMC article. Review.
-
Functions and metabolism of sphingolipids in Saccharomyces cerevisiae.Prog Lipid Res. 2006 Nov;45(6):447-65. doi: 10.1016/j.plipres.2006.03.004. Epub 2006 Apr 21. Prog Lipid Res. 2006. PMID: 16730802 Review.
Cited by
-
Insights into eisosome assembly and organization.J Biosci. 2012 Jun;37(2):295-500. doi: 10.1007/s12038-012-9206-6. J Biosci. 2012. PMID: 22581335 Review.
-
TOR complex 2 is a master regulator of plasma membrane homeostasis.Biochem J. 2022 Sep 30;479(18):1917-1940. doi: 10.1042/BCJ20220388. Biochem J. 2022. PMID: 36149412 Free PMC article.
-
A genome-wide screen for genes affecting eisosomes reveals Nce102 function in sphingolipid signaling.J Cell Biol. 2009 Jun 29;185(7):1227-42. doi: 10.1083/jcb.200811081. J Cell Biol. 2009. PMID: 19564405 Free PMC article.
-
Calcineurin-responsive zinc finger transcription factor CRZ1 of Botrytis cinerea is required for growth, development, and full virulence on bean plants.Eukaryot Cell. 2008 Apr;7(4):584-601. doi: 10.1128/EC.00426-07. Epub 2008 Feb 8. Eukaryot Cell. 2008. PMID: 18263765 Free PMC article.
-
The sphingolipid long-chain base-Pkh1/2-Ypk1/2 signaling pathway regulates eisosome assembly and turnover.J Biol Chem. 2008 Apr 18;283(16):10433-44. doi: 10.1074/jbc.M709972200. Epub 2008 Feb 22. J Biol Chem. 2008. PMID: 18296441 Free PMC article.
References
-
- Audhya, A., and S. D. Emr. 2002. Stt4 PI 4-kinase localizes to the plasma membrane and functions in the Pkc1-mediated MAP kinase cascade. Dev. Cell 2:593-605. - PubMed
-
- Beeler, T., D. Bacikova, K. Gable, L. Hopkins, C. Johnson, H. Slife, and T. Dunn. 1998. The Saccharomyces cerevisiae TSC10/YBR265w gene encoding 3-ketosphinganine reductase is identified in a screen for temperature-sensitive suppressors of the Ca2+-sensitive csg2Delta mutant. J. Biol. Chem. 273:30688-30694. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
