Highly fluorescent noble-metal quantum dots
- PMID: 17105412
- PMCID: PMC2735021
- DOI: 10.1146/annurev.physchem.58.032806.104546
Highly fluorescent noble-metal quantum dots
Abstract
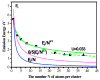
Highly fluorescent, water-soluble, few-atom noble-metal quantum dots have been created that behave as multielectron artificial atoms with discrete, size-tunable electronic transitions throughout the visible and near infrared. These molecular metals exhibit highly polarizable transitions and scale in size according to the simple relation E(Fermi)/N(1/3), predicted by the free-electron model of metallic behavior. This simple scaling indicates that fluorescence arises from intraband transitions of free electrons, and these conduction-electron transitions are the low-number limit of the plasmon-the collective dipole oscillations occurring when a continuous density of states is reached. Providing the missing link between atomic and nanoparticle behavior in noble metals, these emissive, water-soluble Au nanoclusters open new opportunities for biological labels, energy-transfer pairs, and light-emitting sources in nanoscale optoelectronics.
Figures
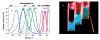




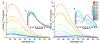

References
-
- Kreibig U, Vollmer M. Optical Properties of Metal Clusters. Berlin: Springer; 1995.
-
- Haberland H. Clusters of Atoms and Molecules: Theory Experiment, and Clusters of Atoms. Berlin: Springer-Verlag; 1994.
-
- El-Sayed MA. Some interesting properties of metals confined in time and nanometer space of different shapes. Accounts of Chemical Research. 2001;34:257. - PubMed
-
- Whetten RL, et al. Crystal structures of molecular gold nanocrystal arrays. Accounts of Chemical Research. 1999;32:397.
-
- Ozin GA, Huber H. Cryophotoclustering Techniques for synthesizing very small naked silver cluster Agn of known size (where n=2–5). The molecular metal cluster-bulk metal particle interface. Inorganic Chemistry. 1978;17:155.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

