Intrinsic ON responses of the retinal OFF pathway are suppressed by the ON pathway
- PMID: 17108159
- PMCID: PMC2553694
- DOI: 10.1523/JNEUROSCI.1718-06.2006
Intrinsic ON responses of the retinal OFF pathway are suppressed by the ON pathway
Abstract
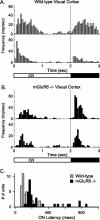
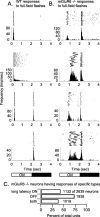
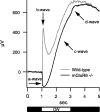
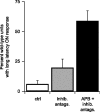
Parallel ON and OFF pathways conduct visual signals from bipolar cells in the retina to higher centers in the brain. ON responses are thought to originate by exclusive use of metabotropic glutamate receptor 6 (mGluR6) expressed in retinal ON bipolar cells. Paradoxically, we find ON responses in retinal ganglion cells of mGluR6-null mice, but they occur at long latency. The long-latency ON responses are not blocked by metabotropic glutamate or cholinergic receptor antagonists and are not produced by activation of receptive field surrounds. We show that these longer-latency ON responses are initiated in the OFF pathways. Our results expose a previously unrecognized intrinsic property of OFF retinal pathways that generates responses to light onset. In mGluR6-null mice, long-latency ON responses are observed in the visual cortex, indicating that they can be conducted reliably to higher visual areas. In wild-type (WT) mice, APB (DL-2-amino-4-phosphonobutyric acid), an mGluR6 agonist, blocks normal, short-latency ON responses but unmasks longer-latency ones. We find that these potentially confusing ON responses in the OFF pathway are actively suppressed in WT mice via two pharmacologically separable retinal circuits that are activated by the ON system in the retina. Consequently, we propose that a major function of the signaling of the ON pathway to the OFF pathway is suppression of these mistimed, and therefore inappropriate, light-evoked responses.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous
