Stromal cell networks regulate lymphocyte entry, migration, and territoriality in lymph nodes
- PMID: 17112751
- PMCID: PMC2692293
- DOI: 10.1016/j.immuni.2006.10.011
Stromal cell networks regulate lymphocyte entry, migration, and territoriality in lymph nodes
Abstract
After entry into lymph nodes (LNs), B cells migrate to follicles, whereas T cells remain in the paracortex, with each lymphocyte type showing apparently random migration within these distinct areas. Other than chemokines, the factors contributing to this spatial segregation and to the observed patterns of lymphocyte movement are poorly characterized. By combining confocal, electron, and intravital microscopy, we showed that the fibroblastic reticular cell network regulated naive T cell access to the paracortex and also supported and defined the limits of T cell movement within this domain, whereas a distinct follicular dendritic cell network similarly served as the substratum for movement of follicular B cells. These results highlight the central role of stromal microanatomy in orchestrating cell migration within the LN.
Figures



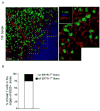
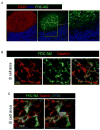

Comment in
-
Rulers over randomness: stroma cells guide lymphocyte migration in lymph nodes.Immunity. 2006 Dec;25(6):867-9. doi: 10.1016/j.immuni.2006.11.002. Immunity. 2006. PMID: 17174930 Review.
References
-
- Anderson AO, Shaw S. T cell adhesion to endothelium: the FRC conduit system and other anatomic and molecular features which facilitate the adhesion cascade in lymph node. Semin Immunol. 1993;5:271–282. - PubMed
-
- Ansel KM, Ngo VN, Hyman PL, Luther SA, Forster R, Sedgwick JD, Browning JL, Lipp M, Cyster JG. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature. 2000;406:309–314. - PubMed
-
- Bousso P, Robey E. Dynamics of CD8+ T cell priming by dendritic cells in intact lymph nodes. Nat Immunol. 2003;4:579–585. - PubMed
-
- Carroll MC. The role of complement in B cell activation and tolerance. Adv Immunol. 2000;74:61–88. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

