Disulfide trapping the mechanosensitive channel MscL into a gating-transition state
- PMID: 17114217
- PMCID: PMC1783893
- DOI: 10.1529/biophysj.106.090316
Disulfide trapping the mechanosensitive channel MscL into a gating-transition state
Abstract
The mechanosensitive channel of large conductance, MscL, serves as a biological emergency release valve protecting bacteria from acute osmotic downshock, and is to date the best characterized mechanosensitive channel. The N-terminal region of the protein has been shown to be critical for function by random, site-directed, and deletion mutagenesis, yet is structurally poorly understood. One model proposes that the extreme N-termini form a cluster of amphipathic helices that serves as a cytoplasmic second gate, separated from the pore-forming transmembrane domain by a "linker". Here, we have utilized cysteine trapping of single-cysteine mutated channels to determine the proximity, within the homopentameric complex, of residues within and just peripheral to this proposed linker. Our results indicate that all residues in this region can form disulfide bridges, and that the percentage of dimers increases when the channel is gated in vivo. Functional studies suggest that oxidation traps one of these mutated channels, N15C, into a gating-transition state that retains the capacity to obtain both fully open and closed states. The data are not easily explained by current models for the smooth transition from closed-to-open states, but predict that an asymmetric movement of one or more of the subunits commonly occurs upon gating.
Figures


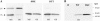
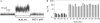

References
-
- Niu, W., and F. Sachs. 2003. Dynamic properties of stretch-activated K+ channels in adult rat atrial myocytes. Prog. Biophys. Mol. Biol. 82:121–135. - PubMed
-
- Tan, J., W. Liu, and D. Saint. 2004. Differential expression of the mechanosensitive potassium channel TREK-1 in epicardial and endocardial myocytes in rat ventricle. Exp. Physiol. 89:237–242. - PubMed
-
- Terrenoire, C., I. Lauritzen, F. Lesage, G. Romey, and M. Lazdunski. 2001. A TREK-1-like potassium channel in atrial cells inhibited by b-adrenergic stimulation and activated by volatile anesthetics. Circ. Res. 89:336–342. - PubMed
-
- Hamill, O. P., and B. Martinac. 2001. Molecular basis of mechanotransduction in living cells. Physiol. Rev. 81:685–740. - PubMed
-
- Sukharev, S., and D. P. Corey. 2004. Mechanosensitive channels: multiplicity of families and gating paradigms. Sci. STKE. 2004:re4. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

