Altered membrane dynamics of quantum dot-conjugated integrins during osteogenic differentiation of human bone marrow derived progenitor cells
- PMID: 17114225
- PMCID: PMC1783865
- DOI: 10.1529/biophysj.106.094896
Altered membrane dynamics of quantum dot-conjugated integrins during osteogenic differentiation of human bone marrow derived progenitor cells
Abstract
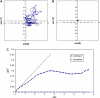
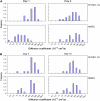
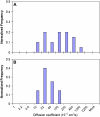
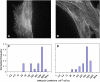
Functionalized quantum dots offer several advantages for tracking the motion of individual molecules on the cell surface, including selective binding, precise optical identification of cell surface molecules, and detailed examination of the molecular motion without photobleaching. We have used quantum dots conjugated with integrin antibodies and performed studies to quantitatively demonstrate changes in the integrin dynamics during osteogenic differentiation of human bone marrow derived progenitor cells (BMPCs). Consistent with the unusually strong BMPC adhesion previously observed, integrins on the surface of undifferentiated BMPC were found in clusters and the lateral diffusion was slow (e.g., approximately 10(-11) cm2/s). At times as early as those after a 3-day incubation in the osteogenic differentiation media, the integrin diffusion coefficients increased by an order of magnitude, and the integrin dynamics became indistinguishable from that measured on the surface of terminally differentiated human osteoblasts. Furthermore, microfilaments in BMPCs consisted of atypically thick bundles of stress fibers that were responsible for restricting the integrin lateral mobility. Studies using laser optical tweezers showed that, unlike fully differentiated osteoblasts, the BMPC cytoskeleton is weakly associated with its cell membrane. Based on these findings, it appears likely that the altered integrin dynamics is correlated with BMPC differentiation and that the integrin lateral mobility is restricted by direct links to microfilaments.
Figures




Similar articles
-
Requirement of alpha(4)beta(1) and alpha(5)beta(1) integrin expression in bone-marrow-derived progenitor cells in preventing endotoxin-induced lung vascular injury and edema in mice.Stem Cells. 2009 Dec;27(12):3112-20. doi: 10.1002/stem.241. Stem Cells. 2009. PMID: 19839056 Free PMC article.
-
Fluid shear-induced mechanical signaling in MC3T3-E1 osteoblasts requires cytoskeleton-integrin interactions.Am J Physiol. 1998 Dec;275(6 Pt 1):C1591-601. Am J Physiol. 1998. PMID: 9843721
-
The integrin alpha9beta1 on hematopoietic stem and progenitor cells: involvement in cell adhesion, proliferation and differentiation.Haematologica. 2009 Nov;94(11):1493-501. doi: 10.3324/haematol.2009.006072. Epub 2009 Jul 16. Haematologica. 2009. PMID: 19608669 Free PMC article.
-
Integrin and cadherin signaling in bone: role and potential therapeutic targets.Trends Endocrinol Metab. 2014 Nov;25(11):567-75. doi: 10.1016/j.tem.2014.06.009. Epub 2014 Jul 14. Trends Endocrinol Metab. 2014. PMID: 25034128 Review.
-
Integrating an integrin: a direct route to actin.Biochim Biophys Acta. 2004 Jul 5;1692(2-3):47-54. doi: 10.1016/j.bbamcr.2004.04.011. Biochim Biophys Acta. 2004. PMID: 15246678 Review.
Cited by
-
Study on intracellular delivery of liposome encapsulated quantum dots using advanced fluorescence microscopy.Sci Rep. 2019 Jul 19;9(1):10504. doi: 10.1038/s41598-019-46732-5. Sci Rep. 2019. PMID: 31324829 Free PMC article.
-
Dynamic partitioning of a glycosyl-phosphatidylinositol-anchored protein in glycosphingolipid-rich microdomains imaged by single-quantum dot tracking.Traffic. 2009 Jun;10(6):691-712. doi: 10.1111/j.1600-0854.2009.00902.x. Epub 2009 Mar 27. Traffic. 2009. PMID: 19416475 Free PMC article.
-
Physicochemical control of adult stem cell differentiation: shedding light on potential molecular mechanisms.J Biomed Biotechnol. 2010;2010:743476. doi: 10.1155/2010/743476. Epub 2010 Apr 1. J Biomed Biotechnol. 2010. PMID: 20379388 Free PMC article. Review.
-
Integrin-adhesion ligand bond formation of preosteoblasts and stem cells in three-dimensional RGD presenting matrices.Biomacromolecules. 2008 Jul;9(7):1843-51. doi: 10.1021/bm8000606. Epub 2008 Jun 10. Biomacromolecules. 2008. PMID: 18540674 Free PMC article.
-
In vivo imaging of embryonic stem cell therapy.Eur J Nucl Med Mol Imaging. 2011 Apr;38(4):774-84. doi: 10.1007/s00259-010-1667-y. Epub 2010 Nov 24. Eur J Nucl Med Mol Imaging. 2011. PMID: 21107558 Review.
References
-
- Cox, E. A., and A. Huttenlocher. 1998. Regulation of integrin-mediated adhesion during cell migration. Microsc. Res. Tech. 43:412–419. - PubMed
-
- Webb, D. J., C. M. Brown, and A. F. Horwitz. 2003. Illuminating adhesion complexes in migrating cells: moving toward a bright future. Curr. Opin. Cell Biol. 15:614–620. - PubMed
-
- Arnaout, M. A., B. Mahalingam, and J. P. Xiong. 2005. Integrin structure, allostery, and bidirectional signaling. Annu. Rev. Cell Dev. Biol. 21:381–410. - PubMed
-
- Juliano, R. L. 2002. Signal transduction by cell adhesion receptors and the cytoskeleton: functions of integrins, cadherins, selectins, and immunoglobulin-superfamily members. Annu. Rev. Pharmacol. Toxicol. 42:283–323. - PubMed
-
- Hynes, R. O. 1992. Integrins: versatility, modulation, and signaling in cell adhesion. Cell. 69:11–25. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

