Mechanism by which MLO-A5 late osteoblasts/early osteocytes mineralize in culture: similarities with mineralization of lamellar bone
- PMID: 17115241
- PMCID: PMC1802097
- DOI: 10.1007/s00223-006-0107-2
Mechanism by which MLO-A5 late osteoblasts/early osteocytes mineralize in culture: similarities with mineralization of lamellar bone
Abstract
The mechanisms whereby bone mineralizes are unclear. To study this process, we used a cell line, MLO-A5, which has highly elevated expression of markers of the late osteoblast such as alkaline phosphatase, bone sialoprotein, parathyroid hormone type 1 receptor, and osteocalcin and will mineralize in sheets, not nodules. In culture, markers of osteocytes and dendricity increase with time, features of differentiation from a late osteoblast to an early osteocyte. Mineral formation was examined using transmission electron microscopy, scanning electron microscopy with energy-dispersive X-ray analysis, and atomic force microscopy. At 3-4 days of culture, spheres of approximately 20-50 nm containing calcium and phosphorus were observed budding from and associated with developing cellular projections. By 5-6 days, these calcified spheres were associated with collagen fibrils, where over time they continued to enlarge and to engulf the collagen network. Coalescence of these mineralized spheres and collagen-mediated mineralization were responsible for the mineralization of the matrix. Similar calcified spheres were observed in cultured fetal rat calvarial cells and in murine lamellar bone. We propose that osteoid-osteocytes generate spherical structures that calcify during the budding process and are fully mineralized on their developing cellular processes. As the cellular process narrows in diameter, these mineralized structures become associated with and initiate collagen-mediated mineralization.
Figures

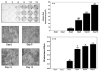
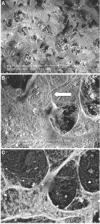
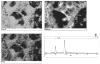





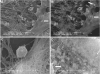


Similar articles
-
Establishment of an osteoid preosteocyte-like cell MLO-A5 that spontaneously mineralizes in culture.J Bone Miner Res. 2001 Sep;16(9):1622-33. doi: 10.1359/jbmr.2001.16.9.1622. J Bone Miner Res. 2001. PMID: 11547831
-
Extracellular matrix mineralization in murine MC3T3-E1 osteoblast cultures: an ultrastructural, compositional and comparative analysis with mouse bone.Bone. 2015 Feb;71:244-56. doi: 10.1016/j.bone.2014.11.003. Epub 2014 Nov 13. Bone. 2015. PMID: 25460184 Free PMC article.
-
Time lapse imaging techniques for comparison of mineralization dynamics in primary murine osteoblasts and the late osteoblast/early osteocyte-like cell line MLO-A5.Cells Tissues Organs. 2009;189(1-4):6-11. doi: 10.1159/000151745. Epub 2008 Aug 27. Cells Tissues Organs. 2009. PMID: 18728354 Free PMC article.
-
The actions of parathyroid hormone on bone: relation to bone remodeling and turnover, calcium homeostasis, and metabolic bone disease. Part I of IV parts: mechanisms of calcium transfer between blood and bone and their cellular basis: morphological and kinetic approaches to bone turnover.Metabolism. 1976 Jul;25(7):809-44. doi: 10.1016/0026-0495(76)90151-7. Metabolism. 1976. PMID: 781470 Review.
-
Cellular Processes by Which Osteoblasts and Osteocytes Control Bone Mineral Deposition and Maturation Revealed by Stage-Specific EphrinB2 Knockdown.Curr Osteoporos Rep. 2019 Oct;17(5):270-280. doi: 10.1007/s11914-019-00524-y. Curr Osteoporos Rep. 2019. PMID: 31401710 Review.
Cited by
-
Osteoblast biocompatibility of premineralized, hexamethylene-1,6-diaminocarboxysulfonate crosslinked chitosan fibers.J Biomed Mater Res A. 2015 Oct;103(10):3201-11. doi: 10.1002/jbm.a.35451. Epub 2015 Mar 30. J Biomed Mater Res A. 2015. PMID: 25771925 Free PMC article.
-
Live Imaging of Type I Collagen Assembly Dynamics in Osteoblasts Stably Expressing GFP and mCherry-Tagged Collagen Constructs.J Bone Miner Res. 2018 Jun;33(6):1166-1182. doi: 10.1002/jbmr.3409. Epub 2018 Mar 23. J Bone Miner Res. 2018. PMID: 29461659 Free PMC article.
-
3D Interrelationship between Osteocyte Network and Forming Mineral during Human Bone Remodeling.Adv Healthc Mater. 2021 Jun;10(12):e2100113. doi: 10.1002/adhm.202100113. Epub 2021 May 8. Adv Healthc Mater. 2021. PMID: 33963821 Free PMC article.
-
Parathyroid Hormone Induces Bone Cell Motility and Loss of Mature Osteocyte Phenotype through L-Calcium Channel Dependent and Independent Mechanisms.PLoS One. 2015 May 5;10(5):e0125731. doi: 10.1371/journal.pone.0125731. eCollection 2015. PLoS One. 2015. PMID: 25942444 Free PMC article.
-
Osteocytes but not osteoblasts directly build mineralized bone structures.Int J Biol Sci. 2021 Jun 11;17(10):2430-2448. doi: 10.7150/ijbs.61012. eCollection 2021. Int J Biol Sci. 2021. PMID: 34326685 Free PMC article.
References
-
- Bordier MP, Miravet L, Ryckerwaert A, Rasmussen H. Morphological and morphometrical characteristics of the mineralization front. A vitamin D regulated sequence of bone remodeling. In: Meunier PJ, editor. Bone Histomorphometry. Armour Montagu; Paris: 1976. pp. 335–354.
-
- Nijweide PJ, van der Plas A, Scherft JP. Biochemical and histological studies on various bone cell preparations. Calcif Tissue Int. 1981;33:529–540. - PubMed
-
- Palumbo C. A three-dimensional ultrastructural study of osteoid-osteocytes in the tibia of chick embryos. Cell Tissue Res. 1986;246:125–131. - PubMed
-
- Owen M. Cell population kinetics of an osteogenic tissue I. Clin Orthop Relat Res. 1995;313:3–7. - PubMed
-
- Mikuni-Takagaki Y, Kakai Y, Satoyoshi M, Kawano E, Suzuki Y, Kawase T, Saito S. Matrix mineralization and the differentiation of osteocyte-like cells in culture. J Bone Miner Res. 1995;10:231–242. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

